Research Articles
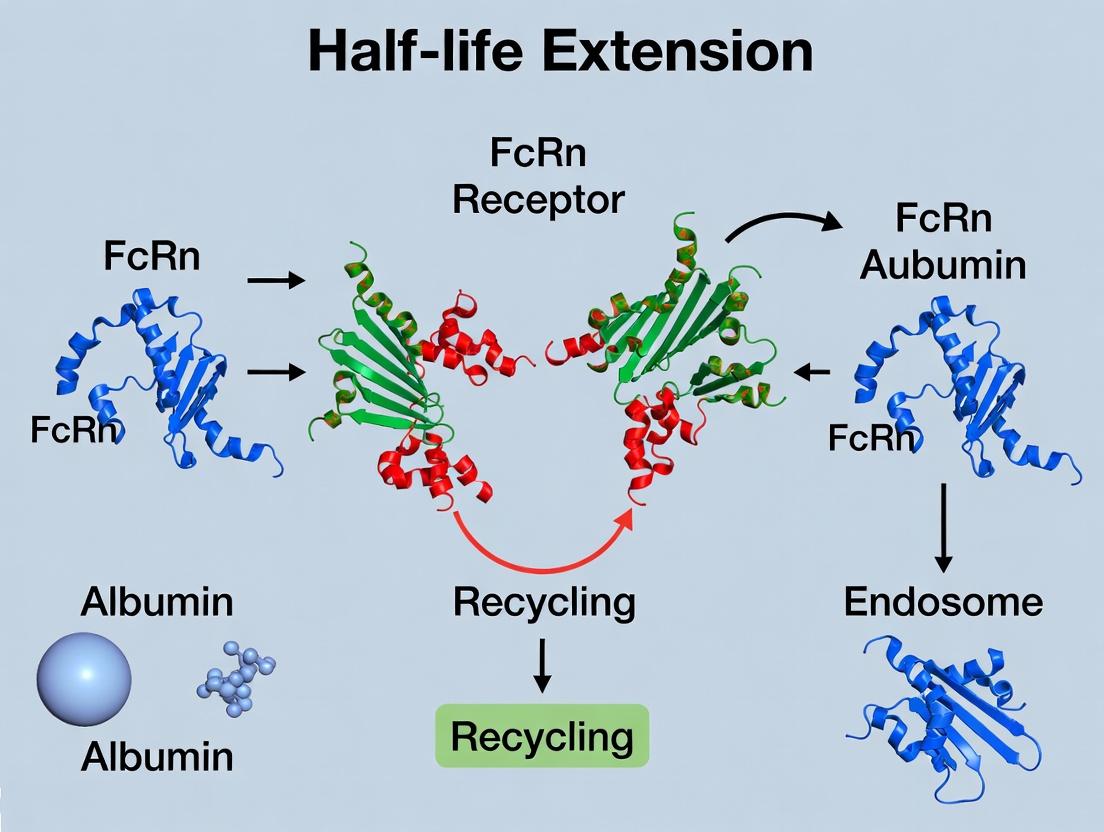
Harnessing the FcRn Receptor: A Comprehensive Guide to Albumin Half-Life Extension for Therapeutic Proteins
This article provides a detailed technical overview of the neonatal Fc receptor (FcRn) for drug development professionals, covering its fundamental biology in IgG and albumin recycling, methodologies for engineering half-life...
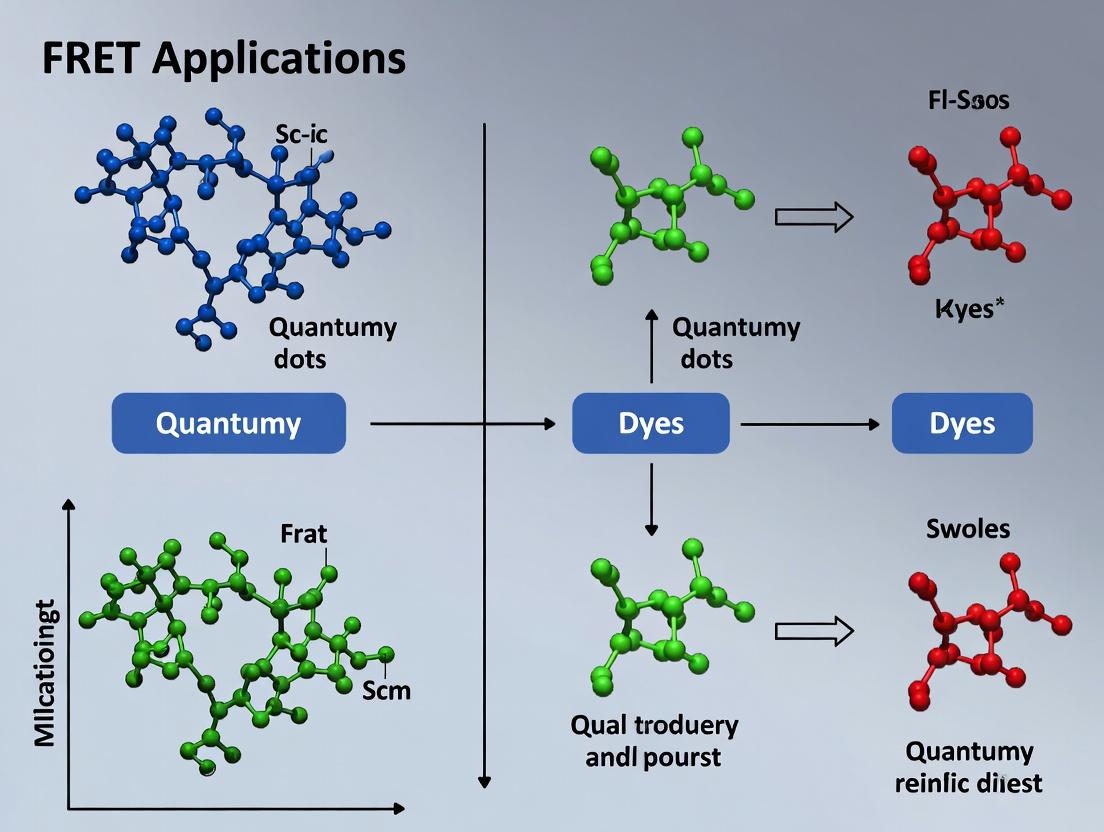
FRET Biosensing Revolution: Leveraging Quantum Dots and Organic Dyes for Advanced Biomedical Research and Drug Discovery
This comprehensive guide explores the transformative role of Förster Resonance Energy Transfer (FRET) in biomedical research, focusing on the synergistic pairing of quantum dots (QDs) as donors with organic dyes...
Navigating the Frontier: A Deep Dive into the FDA's Regulatory Science Research Plan for Nanotechnology
This article provides a comprehensive analysis of the U.S.
Navigating FDA Premarket Review for Nanotech Products: A Comprehensive Guide for Researchers and Developers
This article provides a detailed, current guide for researchers, scientists, and drug development professionals on the FDA's premarket review requirements for nanotechnology-enabled products.
Navigating the Future of Nanomedicine: How the FDA Nanotechnology Task Force Report Transforms Drug Development & Regulation
This comprehensive analysis examines the lasting impact of the FDA's 2022 Nanotechnology Task Force report on biomedical research and pharmaceutical development.
Navigating the FDA's Regulatory Framework for Nanotechnology Drug Products: A 2024 Guide for Researchers and Developers
This comprehensive guide examines the U.S.
Navigating FDA Guidelines for Nanotechnology Drug Development: A Strategic Framework for Industry Success
This article provides a comprehensive roadmap for researchers and drug development professionals engaging with the FDA on nanotechnology-based products.
Navigating FDA Nanomedicine Guidance: Key Requirements for Drug Development Professionals
This comprehensive guide analyzes the FDA's current regulatory framework for drug products containing nanomaterials.
Navigating FDA Guidance for Nanomedicine: A Strategic Framework for Drug Development Success
This article provides a comprehensive overview of the U.S.
FDA and OECD Nanotechnology Guidelines: Aligning Test Protocols for Drug Development Success
This article provides a comprehensive guide for researchers and drug development professionals on the critical alignment between U.S.