Visualizing the Unseen: How SCP-Nano Technology Enables Single-Cell Resolution Imaging for Nanocarrier Tracking in Drug Delivery
This article provides a comprehensive guide to SCP-Nano, an advanced imaging platform enabling single-cell-resolution tracking of nanocarriers.
Visualizing the Unseen: How SCP-Nano Technology Enables Single-Cell Resolution Imaging for Nanocarrier Tracking in Drug Delivery
Abstract
This article provides a comprehensive guide to SCP-Nano, an advanced imaging platform enabling single-cell-resolution tracking of nanocarriers. We explore its foundational principles and technological synergy, detail methodologies for live-cell imaging and biodistribution analysis, address common experimental challenges and optimization strategies, and validate its performance against established techniques. Designed for researchers and drug development professionals, this resource synthesizes current knowledge to empower the precise development and evaluation of next-generation nanomedicines.
Demystifying SCP-Nano: The Core Principles and Evolution of Single-Cell Nanocarrier Imaging
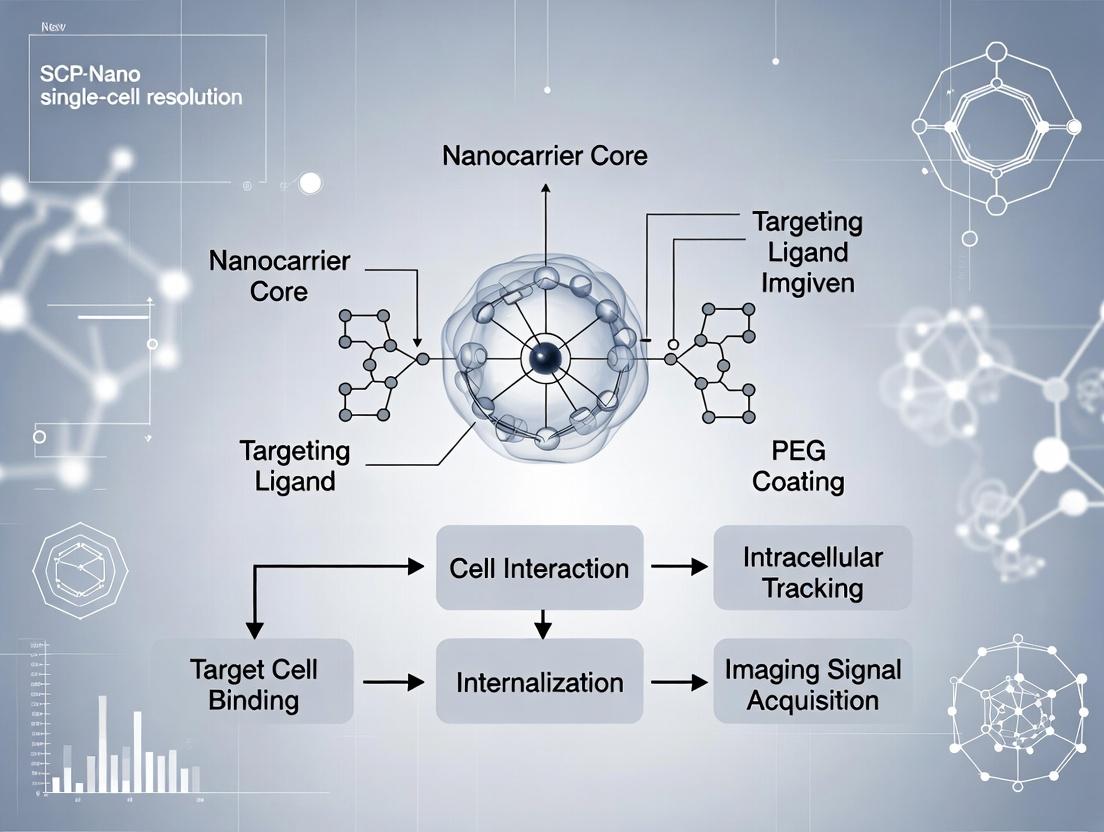
This whitepaper, framed within a broader thesis on single-cell resolution nanocarrier imaging, details the SCP-Nano (Single-Cell Particle Nanocarrier) imaging paradigm. This approach enables the precise tracking, quantification, and functional analysis of individual therapeutic or diagnostic nanoparticles within complex biological systems at the single-cell level, revolutionizing target engagement and pharmacokinetic studies in drug development.
The SCP-Nano paradigm transcends bulk measurements by focusing on the heterogeneity of nanocarrier-cell interactions. It integrates high-resolution microscopy, single-particle tracking (SPT), and multiplexed biosensing to correlate nanocarrier biodistribution with functional cellular outcomes in real time.
Table 1: Comparison of Imaging Modalities for SCP-Nano
| Modality | Spatial Resolution | Temporal Resolution | Key Advantage for SCP-Nano | Primary Limitation |
|---|---|---|---|---|
| Confocal Microscopy | ~200 nm | Seconds | 3D sectioning, multi-label | Photobleaching |
| Total Internal Reflection (TIRF) | ~100 nm | Milliseconds | High signal-to-noise for membrane events | Limited to surface proximity |
| Stimulated Emission Depletion (STED) | ~30 nm | Seconds | Sub-diffraction resolution | Complex setup, high photostress |
| MINFLUX Nanoscopy | ~1-5 nm | Milliseconds | Ultimate localization precision | Specialized instrumentation |
| Correlative Light/Electron Microscopy (CLEM) | ~1 nm (EM) | N/A | Ultrastructural context | Non-live, technically demanding |
Table 2: Quantitative Metrics Derived from SCP-Nano Analysis
| Metric | Description | Typical Value Range (from recent studies) | Significance |
|---|---|---|---|
| Particle Binding Rate (k_on) | Association constant per cell | 0.01 - 0.5 s⁻¹ | Target affinity & avidity |
| Intracellular Trajectory Mean Squared Displacement (MSD) | Measure of particle motility | Anomalous diffusion (α = 0.3-0.7) | Indicates transport mechanism (trapped, vesicular, free) |
| Single-Cell Dose | Number of particles internalized per cell | 1 - 500+ | Heterogeneity in delivery efficiency |
| Lysosomal Escape Fraction | % of particles escaping endo/lysosomes | 5% - 60% | Key for nucleic acid/gene delivery efficacy |
Experimental Protocols
Protocol 3.1: Single-Particle Tracking (SPT) of Targeted Liposomes
Objective: Quantify the binding kinetics and mobility of antibody-conjugated liposomes on live target cells. Materials: See "The Scientist's Toolkit" below. Procedure:
- Nanocarrier Labeling: Incorporate 0.5-1 mol% of a lipophilic dye (e.g., DiD) and 0.1 mol% of a quencher into the lipid bilayer of ~100 nm liposomes. Conjugate targeting ligands (e.g., antibodies) via PEG spacer using click chemistry.
- Cell Preparation: Seed target cells (e.g., HER2+ SK-BR-3) on glass-bottom dishes 24h prior. Before imaging, replace medium with phenol-free imaging buffer.
- Data Acquisition: Using a TIRF or highly inclined illumination microscope, record at 10-100 fps for 5 minutes before and 20 minutes after adding 50-100 pM of labeled liposomes.
- Analysis: Use TrackMate (Fiji) or custom Python scripts to:
- Detect particles using Laplacian of Gaussian filter.
- Link detections into trajectories using a linear motion model.
- Calculate MSD vs. time lag: MSD(τ) = 4Dτ^α. Fit to determine diffusion coefficient (D) and anomalous exponent (α).
Protocol 3.2: Correlative SCP-Nano and Single-Cell RNA Sequencing
Objective: Link nanocarrier fate to transcriptional profiles of the same cell. Procedure:
- Fate-Specific Staining: Treat cells with fluorescently labeled nanoparticles (e.g., siRNA-lipid nanoparticles). After 6-24h, stain with LysoTracker (lysosomes) and Hoechst (nucleus).
- High-Content Imaging: Use an automated confocal microscope to image entire well. Identify and classify cells based on nanoparticle localization (e.g., "lysosomal trapped" vs. "cytoplasmic dispersed").
- Cell Selection & Lysis: Using a micropatterning-compatible platform or laser capture microdissection, isolate specific single cells into individual wells of a 96-well plate containing lysis buffer.
- scRNA-seq Library Prep: Perform reverse transcription and cDNA amplification using a platform-specific kit (e.g., 10x Genomics). Sequence libraries.
- Data Integration: Align sequencing data to cell image classifications to identify gene expression signatures correlating with efficient cytosolic delivery.
Visualization of Key Pathways and Workflows
Title: SCP-Nano Imaging & Analysis Workflow
Title: Intracellular Trafficking Pathways for Nanocarriers
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for SCP-Nano Experiments
| Item | Function in SCP-Nano | Example Product/Catalog Number (Representative) |
|---|---|---|
| Fluorescent Lipophilic Dyes | High-density labeling of lipid-based nanocarriers for single-particle brightness. | DiD, DiI, DiR (Thermo Fisher, D-307, D-282, D-12761) |
| pH-Sensitive Fluorophores | Report on nanocarrier location in acidic compartments (endosomes/lysosomes). | pHrodo dyes (Thermo Fisher, P36600) or CypHer5E. |
| Quantum Dots with PEG Ligands | Extremely bright, photostable inorganic probes for long-duration tracking. | CdSe/ZnS QDs, carboxylic acid-functionalized (Sigma, 900311). |
| Organelle-Specific Live Cell Stains | Contextualize nanoparticle location (lysosomes, mitochondria, ER). | LysoTracker Deep Red, MitoTracker Green (Thermo Fisher, L12492, M7514). |
| Blocking Polymers (e.g., Pluronic F-127) | Reduce non-specific binding of nanoparticles to substrates and cells during imaging. | Pluronic F-127 (Sigma, P2443). |
| Glass-Bottom Culture Dishes | Provide optimal optical clarity for high-resolution, high-NA objective lenses. | MatTek dishes (P35G-1.5-14-C) or Ibidi µ-Dishes (81158). |
| Oxygen Scavenging & Anti-blinking Systems | Prolong fluorophore longevity and reduce blinking in single-molecule studies. | Glucose oxidase/catalase systems or commercial ROXS buffers (e.g., from GattaQuant). |
This whitepaper details the convergence of three cornerstone technologies—super-resolution microscopy (SRM), intravital imaging (IVI), and advanced probe design—as the enabling framework for SCP-Nano (Single-Cell Precision Nanocarrier) research. The overarching thesis posits that only through this synergy can we achieve the requisite spatiotemporal resolution and functional readouts to deconstruct the in vivo journey of therapeutic nanocarriers, from systemic circulation to target-cell engagement and subcellular fate. This guide provides the technical foundation for implementing this integrated approach in preclinical drug development.
Technology Pillars: Principles and Quantitative Comparison
Super-Resolution Microscopy (SRM)
SRM techniques surpass the diffraction limit (~250 nm lateral) of conventional light microscopy. The following table compares key modalities relevant to SCP-Nano imaging.
Table 1: Comparative Analysis of Super-Resolution Microscopy Techniques
| Technique | Principle | Effective Resolution (XY) | Temporal Resolution | Key Advantage for SCP-Nano | Primary Limitation |
|---|---|---|---|---|---|
| STED | Depletes emission periphery via stimulated emission. | 20-80 nm | Milliseconds-Seconds | High resolution in tissue sections; quantifiable. | High laser power; complex implementation. |
| SIM | Reconstructs high-frequency information from patterned illumination. | 100-120 nm | Sub-second | Good speed; compatible with standard fluorophores. | Moderate resolution gain; reconstruction artifacts. |
| SMLM (e.g., PALM/STORM) | Activates sparse subsets of photoswitchable probes for localization. | 10-30 nm | Seconds-Minutes | Highest resolution; molecular counting. | Slow; requires special probes/photochemistry. |
| Expansion Microscopy | Physical sample expansion post-labeling. | ~70 nm (post-expansion) | N/A (fixed samples) | Uses conventional microscopes; achieves ~70 nm resolution. | Requires fixed tissue; expansion heterogeneity. |
Intravital Imaging (IVI)
IVI allows longitudinal observation of biological processes in living organisms. Key quantitative parameters are summarized below.
Table 2: Intravital Imaging Modalities and Parameters
| Modality | Penetration Depth | Temporal Resolution | Spatial Resolution | Best for SCP-Nano Imaging of: |
|---|---|---|---|---|
| Intravital Multiphoton Microscopy | 500-1000 µm | Seconds-Minutes | Sub-micron (diffraction-limited) | Deep tissue (e.g., tumor, liver) dynamics & cellular interactions. |
| Intravital Confocal Microscopy | 50-150 µm | Sub-second | Sub-micron (diffraction-limited) | Superficial structures (e.g., skin, lymph nodes, eye). |
| Light-Sheet Fluorescence Microscopy | Whole organ/embryo | Seconds | 1-5 µm (typically) | High-speed, volumetric tracking in cleared organs or translucent models. |
| Intravital Optical Coherence Tomography | 1-2 mm | Millisecond | 1-15 µm (axial) | Large-scale vascular flow and nanoparticle accumulation. |
Advanced Probes
Probes are the molecular interpreters of the SCP-Nano story. Their properties dictate the signal specificity, brightness, and compatibility with SRM/IVI.
Table 3: Classes of Advanced Probes for Convergent Imaging
| Probe Class | Example Types | Emission Properties | Compatible Modalities | Key Function in SCP-Nano |
|---|---|---|---|---|
| Organic Fluorophores | Alexa Fluor, Cy dyes, Janelia Fluor. | Bright, photostable. | All (SIM, Confocal, MPM). | Nanocarrier surface labeling; vascular integrity. |
| Photoswitchable/Activatable | PA-JF dyes, caged fluorescein. | Turn on/off with light. | SMLM (PALM/STORM). | Tracking single nanocarrier diffusion. |
| Fluorescent Proteins | GFP, mCherry, Dronpa. | Genetically encoded. | All, best for cell-specific labeling. | Reporter for target cell type (e.g., tumor cells). |
| Lanthanide Nanoparticles | Upconversion nanoparticles (UCNPs). | Anti-Stokes shift; no bleaching. | STED, IVI (deep tissue). | Background-free deep-tissue nanocarrier tracking. |
| Biosensors | FRET-based (e.g., Epac-cAMP). | Rationetric signal change. | Confocal, Multiphoton. | Reporting cellular response (e.g., drug release, cAMP). |
Convergent Experimental Protocols
Integrated Protocol: Tracking SCP-Nano Endocytosis and Endosomal Escape In Vivo
This protocol combines intravital multiphoton microscopy with subsequent STED imaging of explanted tissue.
A. Materials & Animal Preparation
- SCP-Nano Formulation: Lipid nanoparticles (LNPs) labeled with a membrane-incorporated lipophilic dye (e.g., DiD, #Ex/Em 644/665 nm) and loaded with a pH-sensitive fluorescent cargo (e.g., pHrodo Red, #Ex/Em 560/585 nm, fluoresces in acidic endosomes).
- Mouse Model: Transgenic mouse with target tissue (e.g., tumor) expressing a cell-specific fluorescent protein (e.g., GFP under a Tie2 promoter for endothelial cells).
- Surgical Preparation: Anesthetize mouse and create a dorsal skinfold window chamber or perform a minimal laparotomy for liver imaging. Maintain physiological temperature and hydration.
B. Intravital Multiphoton Imaging Workflow
- Administration: Inject DiD/pHrodo-loaded SCP-Nanos intravenously.
- Image Acquisition: Using a multiphoton microscope:
- Excitation: 1040 nm laser for simultaneous GFP, DiD, and pHrodo excitation.
- Detection: Use spectral PMTs to collect: Channel 1 (500-550 nm, GFP), Channel 2 (575-610 nm, pHrodo), Channel 3 (650-720 nm, DiD).
- Time Series: Capture z-stacks (every 2 µm) of the target region every 30 seconds for 60 minutes.
- Vital Signs: Monitor heart rate and SpO2 throughout.
C. Tissue Processing for Correlative SRM
- Perfusion & Fixation: At a key time point (e.g., 30 min post-injection), perfuse mouse transcardially with PBS followed by 4% PFA.
- Tissue Sectioning: Excise target tissue, cryoprotect, and prepare 10-20 µm cryosections.
- Immunostaining: Stain for specific organelles (e.g., anti-EEA1 for early endosomes, anti-LAMP1 for lysosomes) using secondary antibodies conjugated to dyes compatible with STED (e.g., Abberior STAR 635).
D. Correlative STED Imaging Protocol
- Locate Region of Interest (ROI): Using the intravital map coordinates, locate the same ROI on the stained section using confocal mode.
- STED Acquisition:
- Use a 640 nm excitation laser and a 775 nm depletion laser (donut mode).
- Acquire sequential channels: DiD (nanocarrier), pHrodo (acidic compartment), STAR 635 (organelle marker).
- Pixel size: 20 nm, dwell time: 10 µs, 5 line accumulations.
- Analysis: Colocalization analysis (Manders' coefficients) of DiD (nanocarrier) with pHrodo and organelle markers quantifies endosomal entrapment vs. escape.
Protocol: SMLM for Single SCP-Nano Receptor Mapping on Cell Membranes
This protocol uses direct STORM (dSTORM) to visualize the nanoscale distribution of receptors bound by SCP-Nanos on fixed cells.
- Sample Preparation: Incubate target cells with SCP-Nanos bearing a targeting ligand (e.g., anti-EGFR) and conjugated to a photoswitchable dye (e.g., Alexa Fluor 647).
- Fixation: Fix cells with 4% PFA + 0.1% glutaraldehyde for 10 min, then quench with 0.1% NaBH₄.
- Imaging Buffer: Use a STORM imaging buffer: 50 mM Tris, 10 mM NaCl, 10% glucose, 0.5 mg/mL glucose oxidase, 40 µg/mL catalase, and 100 mM mercaptoethylamine (MEA) at pH 8.0.
- dSTORM Acquisition:
- Use a TIRF or HILO configuration. Continuously illuminate with a 640 nm laser (2-5 kW/cm²).
- Acquire 15,000-30,000 frames at 50-100 ms exposure.
- Use a 405 nm activation laser at low power (0-5% of max) to maintain a sparse subset of active emitters.
- Localization & Reconstruction: Use software (e.g., ThunderSTORM, Picasso) to detect single-molecule events, fit their PSF, and reconstruct a super-resolution image. Cluster analysis (e.g., DBSCAN) can quantify receptor clustering induced by SCP-Nano binding.
Visualizing Workflows and Pathways
Title: Correlative Intravital and STED Imaging Workflow
Title: SCP-Nano Cellular Trafficking and Fate Pathway
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Reagents for Convergent SCP-Nano Imaging Research
| Reagent/Material | Supplier Examples | Function in SCP-Nano Research |
|---|---|---|
| Lipophilic Tracers (DiD, DiR) | Thermo Fisher, Biotium | Stable integration into nanocarrier lipid membranes for long-term in vivo tracking. |
| pH-Sensitive Dyes (pHrodo, CypHer5E) | Thermo Fisher | Report on nanocarrier localization in acidic compartments (endosomes/lysosomes). |
| Photoswitchable Dyes (PA-JF549, Alexa Fluor 647) | Janelia, Thermo Fisher | Enable SMLM for nanoscale mapping of nanocarrier binding sites. |
| STED-Compatible Secondary Antibodies (Abberior STAR, Alexa Fluor 594) | Abberior, Thermo Fisher | Allow correlative SRM of nanocarriers with subcellular structures. |
| Oxygen Scavenging / STORM Buffers (Glox/Catalase, MEA) | Sigma-Aldrich, Gattaquant | Enable single-molecule switching for dSTORM imaging of nanocarriers. |
| Genetically Encoded Biosensors (cAMP, Ca2+) | Addgene, Thermo Fisher | Monitor intracellular therapeutic response upon SCP-Nano payload delivery. |
| Dorsal Skinfold Window Chamber | Various custom fabricators | Enables stable, longitudinal intravital imaging of tumors or vasculature. |
| Fiducial Markers (TetraSpeck beads) | Thermo Fisher | Facilitate correlation between light microscopy and EM or between IVI and SRM. |
| Tissue Clearing Kits (CUBIC, iDISCO) | Cosmo Bio, Miltenyi Biotec | Enable light-sheet imaging of whole organs for SCP-Nano distribution studies. |
| Anti-PEG or Anti-Polymer Antibodies | Various (e.g., Academia) | Specific detection of PEGylated or polymeric nanocarriers in tissue sections. |
Why Single-Cell Resolution is a Game-Changer for Nanomedicine and Pharmacokinetics
This whitepaper, framed within the SCP-Nano (Single-Cell Profiling Nanocarrier) research thesis, argues that achieving single-cell resolution in nanomedicine is a paradigm shift. It moves the field beyond bulk tissue averages to a precise understanding of intercellular heterogeneity in drug delivery, pharmacokinetics (PK), and pharmacodynamics (PD). This resolution is critical for optimizing next-generation nanocarriers, predicting efficacy, and minimizing off-target toxicity.
The Heterogeneity Challenge: Beyond Bulk Averages
Traditional PK/PD models treat tissues as homogeneous compartments. Single-cell technologies reveal vast heterogeneity in cell state, receptor expression, and endocytic capacity, which directly dictates nanocarrier fate. Bulk measurements mask critical subpopulations that dominate drug uptake, resistance, or toxicity.
Table 1: Impact of Cellular Heterogeneity on Nanocarrier Pharmacokinetics
| Cellular Variable | Impact on Nanocarrier PK | Bulk Measurement Limitation |
|---|---|---|
| Target Antigen Density (e.g., HER2, CD19) | Drives binding affinity and specificity; low-expression cells are missed. | Reports mean expression, obscuring therapeutically relevant low/high subsets. |
| Endocytic Rate | Determines internalization efficiency and intracellular drug release. | Averages fast and slow endocytic cells, misrepresenting delivery kinetics. |
| Immune Cell Phenotype (TAMs, Kupffer cells) | Governs clearance and off-target accumulation in liver/spleen. | Cannot resolve pro-inflammatory vs. anti-inflammatory phagocytic activity. |
| Stromal Cell Interactions | Modifies tumor penetration and binding site barrier effects. | Fails to delineate contribution of cancer-associated fibroblasts vs. tumor cells. |
| Efflux Pump Activity (e.g., P-gp) | Causes sub-therapeutic intracellular drug levels despite delivery. | Obscures identification of resistant cell clones responsible for relapse. |
Key Technological Enablers for Single-Cell PK/PD
The SCP-Nano thesis integrates advanced imaging and omics tools to deconvolute this complexity.
Experimental Protocol 1: High-Parameter Cytometry of Time (CyTOF) for Nanocarrier Cell Association
Aim: To quantify cell-type-specific association of lanthanide-tagged nanocarriers with 40+ simultaneous cellular markers.
- Nanocarrier Synthesis: Conjugate polymer-coated nanocarriers with DOTA chelators loaded with a stable heavy metal isotope (e.g., 141Pr).
- In Vivo Dosing: Administer tagged nanocarriers intravenously to tumor-bearing mouse models.
- Tissue Processing: At defined timepoints (1h, 6h, 24h), harvest organs, create single-cell suspensions.
- Staining: Stain cells with a cocktail of metal-tagged antibodies targeting cell surface markers (immune, endothelial, tumor).
- Acquisition & Analysis: Analyze cells on a CyTOF mass cytometer. The 141Pr signal quantifies nanocarrier association per cell, correlated with deep immunophenotyping.
Diagram Title: CyTOF Workflow for Single-Cell Nanocarrier Tracking
Experimental Protocol 2: Multiplexed Ion Beam Imaging (MIBI) for Spatial PK
Aim: To visualize nanocarrier distribution and co-localization with cellular targets in intact tissue architecture.
- Sample Preparation: Administer nanocarriers tagged with a rare earth metal (e.g., Terbium). Flash-freeze tissues, section.
- Antibody Staining: Stain tissue sections with a panel of primary antibodies, followed by secondary antibodies conjugated to unique metal isotopes.
- MIBI Imaging: The sample is raster-scanned with a focused primary ion beam, releasing secondary ions from both antibody tags and the nanocarrier tag.
- Data Reconstruction: Mass spectrometry reconstructs the spatial distribution of all targets at subcellular resolution (~200 nm).
- Analysis: Quantify nanocarrier signal within phenotypically defined regions (e.g., PD-L1+ tumor cells, CD31+ vasculature).
Diagram Title: Multiplexed Ion Beam Imaging (MIBI) Protocol
The Scientist's Toolkit: Key Research Reagent Solutions
| Tool/Reagent | Function in SCP-Nano Research | Key Consideration |
|---|---|---|
| Elemental Tags (e.g., 141Pr, 159Tb) | Stably incorporate into nanocarriers for detection by mass cytometry/spectroscopy without signal overlap or quenching. | Must not alter nanocarrier surface properties or PK. |
| Mass Cytometry Antibody Panels | Pre-conjugated, titrated antibodies for deep immunophenotyping alongside nanocarrier detection. | Panel design must include lineage markers and targets of therapeutic interest. |
| CODEX/IMC Antibody Panels | Validated antibody conjugates for multiplexed tissue imaging, compatible with metal or fluorescent barcoding. | Tissue fixation and antigen retrieval are critical for performance. |
| Single-Cell RNA Sequencing Kits (10x Genomics) | Profile transcriptional states of cells that have internalized nanocarriers (via sort-seq). | Enables linking of PK outcomes to cellular functional states. |
| Isotopically-Labeled Lipids/Polymers | Synthesize nanocarriers with incorporated 13C/2H for tracking via NanoSIMS or Raman microscopy. | Provides ultimate subcellular resolution for drug distribution. |
| In Vivo Imaging Agents (CT, PET) | Label nanocarriers with iohexol or 89Zr for whole-body, longitudinal tracking to guide single-cell endpoint analysis. | Bridges macro-scale PK with single-cell resolution endpoints. |
Data Integration and Computational Analysis
The SCP-Nano framework requires new computational pipelines.
Table 2: Computational Methods for Single-Cell PK Data Integration
| Method | Application | Output |
|---|---|---|
| Dimensionality Reduction (UMAP, t-SNE) | Visualize high-dimensional CyTOF data, cluster cells by phenotype and nanocarrier uptake. | Identification of outlier subpopulations with extreme uptake/clearance. |
| Spatial Mapping (CellProfiler, Halolink) | Quantify nanocarrier signal within histology-defined tissue compartments from MIBI/IMC data. | Spatial PK metrics: penetration depth, target occupancy, stromal sequestration. |
| Trajectory Inference (PAGA, Monocle3) | Model cellular state transitions influenced by nanocarrier-delivered drug from scRNA-seq data. | Predicts differentiation or resistance pathways activated post-treatment. |
| Pharmacokinetic Modeling (mlrMCP) | Build multiscale models where single-cell data informs cellular rate constants. | Refined PK/PD models with heterogeneity-driven parameters. |
Implications for Drug Development
- Lead Optimization: Select nanocarrier designs based on their ability to target therapeutically relevant cell subsets, not just bulk tumor accumulation.
- Biomarker Discovery: Identify rare cell phenotypes predictive of response or toxicity.
- Clinical Trial Design: Use single-cell PK insights to stratify patients and select rational combination therapies.
- Toxicology: Identify specific off-target cell types responsible for adverse effects at unprecedented resolution.
Integrating single-cell resolution tools into nanomedicine, as championed by the SCP-Nano thesis, transforms pharmacokinetics from a phenomenological science into a mechanistic, predictive framework. By mapping the fate of nanocarriers onto the cellular atlas of living systems, we can engineer smarter therapies, overcome heterogeneity-driven resistance, and usher in an era of truly personalized nanomedicine.
The evolution from bulk tissue analysis to single-cell investigation represents a paradigm shift in biomedical sciences, particularly in drug delivery. This transition is fundamentally enabling the core thesis of SCP-Nano (Single-Cell Profiling via Nanocarriers) research: to map, with nanoscale precision, the heterogeneous cellular responses to therapeutic agents. Where bulk methods averaged signals across millions of cells, masking critical minority populations and dynamic interactions, single-cell technologies now expose this complexity. SCP-Nano leverages this resolution by integrating advanced nanocarriers with high-dimensional imaging and omics, allowing researchers to visualize drug transport, uptake, and intracellular fate at the level of individual cells within a tissue microenvironment. This whitepaper details the technical journey from bulk to single-cell analysis, providing methodologies and frameworks central to this frontier.
The Limitations of Bulk Analysis: A Quantitative Perspective
Bulk tissue analysis, encompassing techniques like whole-tissue homogenization for western blotting, PCR, or mass spectrometry, provides population-averaged data. The critical limitations that single-cell methods overcome are quantitatively summarized below.
Table 1: Key Limitations of Bulk Tissue Analysis in Drug Delivery Studies
| Limitation Parameter | Typical Bulk Measurement | Impact on Drug Delivery Research | Quantitative Example |
|---|---|---|---|
| Cellular Heterogeneity Masking | Averaged signal across all cell types. | Inability to identify which specific cell type (e.g., cancer stem cell vs. differentiated tumor cell) takes up or responds to a nanocarrier. | A 10x higher drug uptake in a rare 5% cell subpopulation is diluted to a mere 1.5x increase in bulk signal. |
| Rare Cell Population Obscuration | Detection threshold ~1% of total population. | Critical responders (e.g., metastatic precursors) or off-target cells are missed. | A 0.1% population causing drug resistance is undetectable. |
| Spatial Context Loss | No inherent spatial information. | Cannot discern if nanocarrier delivery is perivascular, peri-necrotic, or uniform, crucial for understanding penetration barriers. | N/A – information destroyed during homogenization. |
| Dynamic Range Compression | Measured as mean ± standard deviation. | Subpopulation-specific extreme responses (e.g., complete lysosomal entrapment in macrophages vs. cytosolic release in parenchymal cells) are lost. | Bimodal distribution appears as a broad unimodal distribution. |
The Single-Cell Frontier: Core Technologies Enabling SCP-Nano
The single-cell frontier is built upon complementary technological pillars that resolve the limitations in Table 1.
Single-Cell Omics (scOmics)
- scRNA-seq (Single-Cell RNA Sequencing): Reveals transcriptional states of individual cells, identifying targetable pathways and response signatures post-nanocarrier treatment.
- scATAC-seq (Assay for Transposase-Accessible Chromatin): Maps open chromatin regions, informing on epigenetic heterogeneity affecting drug response.
- CITE-seq / REAP-seq: Combines transcriptional profiling with surface protein detection, allowing immunophenotyping alongside functional genomics.
High-Plex Spatial Transcriptomics & Proteomics
- Technologies: NanoString GeoMx DSP, 10x Visium, multiplexed ion beam imaging (MIBI), CODEX.
- Application: Preserves tissue architecture while quantifying 50-100+ RNA or protein targets in situ. For SCP-Nano, this links nanocarrier location (via fluorescent or metal tags) to local cellular phenotypes.
High-Resolution Live-Cell & Intravital Imaging
- Super-Resolution Microscopy (STED, PALM/STORM): Visualizes nanocarrier interaction with subcellular organelles (e.g., endosomal membranes, nuclear pores) at <100 nm resolution.
- Intravital Microscopy (IVM): Tracks real-time nanocarrier behavior in living animal models, capturing dynamics in blood flow, extravasation, and cell-cell transfer.
Experimental Protocols for SCP-Nano Relevant Single-Cell Studies
Protocol 4.1: Multiplexed Imaging of Nanocarrier Uptake and Cell Phenotype (CODEX)
Objective: To spatially map the uptake of a fluorescently-labeled nanocarrier across 30+ cell phenotypes in a fixed tissue section. Materials: See "The Scientist's Toolkit" below. Workflow:
- Tissue Preparation: Flash-freeze or OCT-embed tissue from nanocarrier-dosed model. Cryosection at 5-10 µm thickness.
- CODEX Staining: Conjugate antibodies for phenotypic markers (CD45, EpCAM, α-SMA, etc.) with unique oligonucleotide barcodes (Assignee Reporter). Incubate on tissue sections alongside a direct fluorophore-conjugated nanocarrier or an antibody against its component.
- Cyclic Imaging: a. Stain with all fluorescent Assigner oligos that bind to the Reporter barcodes. b. Image on a customized epifluorescence microscope (e.g., Keyence BZ-X800 with motorized filter cubes). Acquire nanocarrier channel in every cycle. c. Chemically cleave fluorescent signal. d. Repeat cycles (Steps a-c) for all antibody markers (typically 20-50 cycles).
- Data Processing & Analysis: Use CODEX Instrument Software to align cycles and generate single-cell segmentation masks based on nuclear (DAPI) and membrane markers. Extract mean fluorescence intensity (MFI) for the nanocarrier channel and all phenotypic markers for each cell. Perform dimensionality reduction (t-SNE, UMAP) and clustering to identify cell populations with high/low nanocarrier association.
Diagram 1: CODEX Workflow for Spatial Single-Cell Uptake
Protocol 4.2: scRNA-seq of Nanocarrier-Treated Cells Using Hashtag Oligos (HTO)
Objective: To profile the transcriptional response to nanocarrier treatment while pooling multiple conditions (e.g., different formulations, time points) in a single sequencing run, controlling for batch effects. Materials: See "The Scientist's Toolkit" below. Workflow:
- Sample Preparation & HTO Labeling: Treat cell populations (e.g., primary tumor cell mix) with different nanocarrier formulations (A, B, C) and an untreated control (D). Dissociate into single-cell suspensions. Label each condition's cells with a unique lipid-anchored Hashtag Oligonucleotide (HTO, e.g., BioLegend TotalSeq-A).
- Cell Pooling: Combine all HTO-labeled cell suspensions into one pool.
- Single-Cell Library Prep: Process the pooled sample through a standard 10x Chromium Next GEM workflow. This generates two libraries: the Gene Expression (GEX) library and the Feature Barcode (HTO) library.
- Sequencing & Demultiplexing: Sequence both libraries. Use Cell Ranger (10x Genomics) or Seurat to align GEX reads and count HTO molecules per cell.
- Doublet Detection & Assignment: Apply algorithms (e.g.,
HTODemuxin Seurat) to assign each cell to its original sample (A, B, C, D) based on HTO counts. Remove doublets (cells positive for multiple HTOs). - Differential Expression Analysis: Compare gene expression profiles between conditions (e.g., Nanocarrier A vs. Control D) to identify formulation-specific pathways, stress responses, or efficacy signatures.
Diagram 2: Hashtag Oligo scRNA-seq for Pooled Conditions
The Scientist's Toolkit: Key Reagents & Materials
Table 2: Essential Research Reagent Solutions for SCP-Nano Single-Cell Studies
| Item | Supplier Examples | Function in SCP-Nano Research |
|---|---|---|
| TotalSeq Antibodies (Hashtag & Protein) | BioLegend | For multiplexed sample indexing (HTOs) and surface protein detection alongside scRNA-seq in the same cell. |
| CODEX Antibody Conjugation Kits & Instrument | Akoya Biosciences | For conjugating antibodies with DNA barcodes and performing high-plex cyclic imaging on tissue sections. |
| 10x Chromium Next GEM Kits | 10x Genomics | For partitioning single cells and generating barcoded cDNA libraries for scRNA-seq, scATAC-seq. |
| Cell Hashtag Oligos (HTOs) | Custom Synthesis (IDT) / BioLegend | Sample-specific barcodes for pooling multiple conditions in one scRNA-seq run, reducing cost and batch effects. |
| Live-Cell Organelle Trackers | Thermo Fisher, Abcam | Fluorescent dyes (LysoTracker, MitoTracker) to colocalize with labeled nanocarriers in live-cell imaging assays. |
| CITE-seq Antibody Panels | ProteinCell, BioLegend | Pre-designed panels of oligo-conjugated antibodies for simultaneous protein and RNA measurement at single-cell level. |
| Visium Spatial Gene Expression Slides | 10x Genomics | For capturing whole transcriptome data from tissue sections while retaining spatial location information. |
| Metal-Labeled Antibodies for Imaging Mass Cytometry | Standard BioTools | Antibodies conjugated to rare earth metals for use with Hyperion/IMC to detect 40+ proteins simultaneously in tissue. |
Data Integration & The SCP-Nano Thesis: A Pathway View
The ultimate power of single-cell analysis lies in integrating multimodal data. The SCP-Nano thesis proposes a pipeline where spatial nanocarrier imaging (Protocol 4.1) informs cell sorting or region selection for deep scRNA-seq profiling (Protocol 4.2). This integrated data reveals the signaling networks activated specifically in high-uptake versus low-uptake cells.
Diagram 3: SCP-Nano Data Integration for Target Discovery
Integrated Signaling Pathway Analysis: From the integrated data, a pathway emerges. For example, in tumor-associated macrophages (TAMs) showing high nanocarrier uptake (from CODEX data), scRNA-seq may reveal upregulation of the LRP1 receptor and downstream MEK/ERK and PI3K/Akt survival pathways. Conversely, TAMs with low uptake may show an IRF/STAT1 inflammatory pathway. This provides a mechanistic basis for designing nanocarriers that actively target LRP1 to modulate specific intracellular pathways in a desired cell population.
The frontier of drug delivery has irrevocably shifted to the single-cell level. By moving beyond the averaging obscurity of bulk analysis, SCP-Nano and related frameworks empower researchers to deconvolute the complex interplay between nanocarrier design, heterogeneous cellular uptake, and precise biological outcomes. The experimental protocols and tools detailed herein provide a roadmap for conducting rigorous single-cell resolution studies, ultimately accelerating the rational design of more effective, targeted, and personalized therapeutic delivery systems.
Key Physical and Optical Principles Underpinning SCP-Nano Resolution and Sensitivity
Within the broader thesis on SCP-Nano (Single-Cell Profiling via Nanocarrier Imaging) research, achieving high-resolution, sensitive detection of biomolecules within individual cells is paramount. This technical guide details the core physical and optical principles that enable the nanoscale resolution and exquisite sensitivity of the SCP-Nano platform. The method hinges on the synthesis of functionalized, optically active nanocarriers, their targeted delivery, and the subsequent detection of their unique signals against the cellular background.
Core Physical Principles
The resolution and sensitivity of SCP-Nano are governed by fundamental physical constraints and engineered solutions.
2.1 Diffraction Limit and Super-Resolution Strategies Conventional optical microscopy is limited by diffraction to approximately 200-250 nm laterally. SCP-Nano bypasses this not by imaging the biomolecule directly, but by localizing discrete, point-source nanocarriers. The effective resolution is determined by the localization precision of the carrier's signal, which can reach sub-10 nm under optimal conditions. This is a form of pointillistic or localization microscopy principle.
2.2 Signal-to-Noise Ratio (SNR) and Sensitivity
Sensitivity is defined as the minimum number of target molecules detectable per cell. It is fundamentally governed by the SNR:
SNR = (I_signal) / σ_noise
where I_signal is the intensity from the nanocarrier and σ_noise is the standard deviation of the background and instrumental noise. SCP-Nano enhances SNR through:
- High-Emission Nanocarriers: Using materials with high quantum yield and absorption cross-sections.
- Spectral Isolation: Employing narrow, stable emission bands distinct from cellular autofluorescence.
- Temporal Gating: Exploiting long-lived luminescence to separate signal from short-lived background fluorescence.
2.3 Binding Kinetics and Labeling Density For accurate quantification, the labeling efficiency—the percentage of target biomolecules bound by a nanocarrier—must be characterized. This depends on the affinity (K_D) of the targeting ligand, nanocarrier valency, and steric accessibility of the epitope. Under-sampling leads to underestimation of molecule count.
Core Optical Principles & Modalities
SCP-Nano employs specific optical phenomena to generate detectable signals.
3.1 Plasmon-Enhanced Luminescence Metal-based nanocarriers (e.g., gold nanostars) support localized surface plasmon resonances (LSPRs). At the plasmonic "hot spots," the electromagnetic field is dramatically enhanced, leading to:
- Amplification of Raman scattering signals (SERS).
- Enhancement of fluorescence from adjacent dyes.
3.2 Upconversion Luminescence (UCL) Lanthanide-doped upconversion nanoparticles (UCNPs) absorb multiple low-energy photons (typically near-infrared, NIR) and emit a single higher-energy photon (visible). This process provides:
- Zero Autofluorescence: Biological samples exhibit negligible NIR-excited UCL.
- Deep Tissue Penetration: NIR excitation scatters less and is less damaging.
3.3 Time-Gated Detection This technique capitalizes on the long photoluminescence lifetimes (µs to ms) of certain nanomaterials (e.g., UCNPs, some lanthanide complexes). By introducing a delay between excitation and detection, short-lived background fluorescence (ns lifetime) is excluded, drastically improving contrast.
Table 1: Comparison of Nanocarrier Optical Properties for SCP-Nano
| Nanocarrier Type | Core Material | Excitation (nm) | Emission (nm) | Quantum Yield | Lifetime | Key Advantage for SCP-Nano |
|---|---|---|---|---|---|---|
| Polymeric Dot | Encapsulated organic dyes | 488, 555 | 520, 570 | 0.6 - 0.9 | ~4 ns | Bright, tunable, biocompatible |
| Upconversion NP | NaYF₄:Yb³⁺,Er³⁺ | 980 (NIR) | 540, 660 | 0.01 - 0.3 | 100 µs - 1 ms | No autofluorescence, deep penetration |
| Surface-Enhanced Raman Scattering (SERS) Tag | Au/Ag core, Raman reporter | 633, 785 | Raman Shift | N/A (Scattering) | <1 ns | Multiplexing (narrow bands), photostable |
| Lanthanide Complex | Eu³⁺, Tb³⁺ chelate | UV (~340) | 615, 545 | 0.1 - 0.5 | 0.1 - 2 ms | Ideal for time-gated detection |
Table 2: Typical SCP-Nano Performance Metrics
| Performance Parameter | Typical Range | Influencing Factors |
|---|---|---|
| Spatial Localization Precision | 5 - 30 nm | Photon count, background, camera pixel size |
| Single-Cell Detection Sensitivity | 10 - 100 copies/cell | Nanocarrier brightness, nonspecific binding |
| Multiplexing Capacity | 5 - 10 targets/assay | Spectral overlap of nanocarrier emissions |
| Labeling Efficiency | 30% - 80% | Ligand affinity, nanocarrier size, epitope accessibility |
Key Experimental Protocols
5.1 Protocol: SCP-Nano Workflow for Single-Cell Surface Protein Profiling
Objective: To quantify the expression level of 5 distinct surface proteins on individual fixed cells. Materials: See "The Scientist's Toolkit" below. Workflow:
- Cell Preparation: Seed cells on #1.5 coverslips. At ~70% confluency, fix with 4% PFA for 15 min at RT. Permeabilize with 0.1% Triton X-100 (if intracellular targets are included). Block with 3% BSA/PBS for 1 hour.
- Primary Antibody Incubation: Incubate with a cocktail of target-specific primary antibodies (conjugated to unique DNA oligo barcodes) in blocking buffer for 2 hours at RT.
- Washing: Wash 3x with PBS + 0.05% Tween-20 (PBS-T).
- Nanocarrier Hybridization: Incubate with a cocktail of complementary DNA-conjugated nanocarriers (e.g., 5 distinct UCNPs). Each nanocarrier type corresponds to one DNA barcode/target. Hybridize for 1.5 hours at 37°C in a humidified chamber.
- Stringency Washes: Perform 2x SSCT (Saline-Sodium Citrate with 0.05% Tween-20) washes at 37°C to remove imperfectly hybridized carriers, followed by 2x PBS washes.
- Mounting: Mount coverslip on slide with antifade mounting medium.
- Imaging: Image on a custom-modified epifluorescence or confocal microscope equipped with:
- A 980 nm laser for UCNP excitation.
- Time-gated detection capabilities (e.g., a pulsed laser and delay generator).
- EMCCD or sCMOS camera.
- Spectral filters to separate emission bands.
- Image Analysis: Use software (e.g., custom MATLAB script, ImageJ) for:
- Nanocarrier Identification & Localization: Apply a bandpass filter and maximum-finder algorithm.
- Spectral Unmixing: If emissions overlap, use linear unmixing based on reference spectra.
- Quantification: Count localized spots per cell (segmented via a membrane stain).
5.2 Protocol: Measuring Localization Precision
Objective: To empirically determine the localization precision of a given nanocarrier type under standard imaging conditions. Workflow:
- Immobilize sparse, non-aggregated nanocarriers on a clean glass slide.
- Acquire a time-lapse movie of 1000 frames under constant illumination.
- For a single, isolated nanocarrier in each frame, fit its Point Spread Function (PSF) with a 2D Gaussian function to determine its center coordinates (xi, yi).
- Calculate the standard deviation of the positions over all frames:
σ_x = std({x_i}), σ_y = std({y_i}). - The experimental localization precision is given by:
Precision = sqrt(σ_x² + σ_y²).
Visualizations
SCP-Nano Assay Workflow
Factors Governing SCP-Nano SNR
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for SCP-Nano Experiments
| Reagent/Material | Function & Role in SCP-Nano | Example Product/Type |
|---|---|---|
| DNA-Barcoded Primary Antibodies | Provides target specificity and a unique oligonucleotide "barcode" for subsequent nanocarrier hybridization. Enables high-level multiplexing. | Custom-conjugated antibodies (e.g., via amine-to-thiol coupling of oligos). |
| Functionalized Optical Nanocarriers | The core detection probe. Must have strong, stable optical signature and surface chemistry for DNA conjugation. | Carboxyl- or streptavidin-coated UCNPs, polymer dots, SERS tags. |
| Complementary DNA Oligonucleotides | Conjugated to nanocarriers; binds to antibody barcodes via Watson-Crick base pairing. Sequence design is critical for specificity. | HPLC-purified, thiol- or amine-modified DNA oligos. |
| Stringency Wash Buffer (e.g., SSCT) | Removes nonspecifically bound or imperfectly hybridized nanocarriers, reducing background. Critical for sensitivity. | Saline-sodium citrate (SSC) buffer with Tween-20 detergent. |
| Time-Gated Imaging System | Microscope equipped to exploit long-lifetime signals. Eliminates short-lived autofluorescence. | System with pulsed laser (e.g., 980nm diode), delay generator, and gated camera. |
| Antifade Mounting Medium | Preserves fluorescence/luminescence signal during imaging. Especially important for long acquisition times. | Commercial medium with radical scavengers (e.g., ProLong Diamond, Mowiol-based). |
| Cell Segmentation Stain | A generic label to define cell boundaries for single-cell analysis. | CellMask membrane stains, cytosolic dyes (e.g., Calcein AM), or nuclear stains (DAPI). |
A Practical Guide to Implementing SCP-Nano Imaging in Nanocarrier Research and Development
This technical guide details standardized protocols for preparing and labeling nanocarriers and cells, a foundational pillar for the broader thesis on SCP-Nano (Single-Cell Profiling via Nanocarriers) research. The objective is to achieve high-fidelity, single-cell resolution imaging to track nanocarrier-cell interactions, internalization pathways, and intracellular fate. Reproducible sample preparation and precise labeling are critical for generating quantitative, biologically relevant data in drug delivery system evaluation.
Nanocarrier Preparation & Labeling Strategies
Nanocarriers (e.g., liposomes, polymeric NPs, dendrimers) require labeling for visualization without altering physicochemical or biological properties.
2.1 Core Labeling Methodologies
| Strategy | Typical Tags/Probes | Conjugation Chemistry | Key Advantage | Potential Drawback |
|---|---|---|---|---|
| Covalent Integration | Cy5, Alexa Fluor 647, ATTO 488 | NHS-ester with amine groups, Maleimide with thiols | Stable, permanent label; controlled dye-to-particle ratio. | Risk of altering surface chemistry and biological interactions. |
| Lipid/Polymer Incorporation | DiD, DiI, DiO, BODIPY | Physically encapsulated or inserted into bilayer/matrix | Minimal surface alteration; suitable for hydrophobic cores. | Dye leakage can lead to false-positive signals. |
| Quantum Dot Encapsulation | CdSe/ZnS QDs | Encapsulation during synthesis or surface conjugation | High brightness, photostability, multiplexing via size. | Potential cytotoxicity; larger size; blinking. |
| Metallic NP Core | Gold, Silver NPs | N/A (inherent signal) | No leaching; enables dark-field, SERS, photoacoustic imaging. | Not fluorescent (unless functionalized); may interfere with therapy. |
2.2 Step-by-Step Protocol: Liposome Labeling via Lipid Incorporation This protocol details labeling of PEGylated liposomes for SCP-Nano imaging.
- Materials: DOPC, Cholesterol, DSPE-PEG2000, fluorescent lipid (e.g., DiD chloroform solution), rotary evaporator, extruder with 100 nm membrane, PBS.
- Procedure: a. Formulation: In a glass vial, mix lipid stocks in chloroform: DOPC (62 mol%), Cholesterol (33 mol%), DSPE-PEG2000 (4 mol%), DiD (0.5-1 mol%). b. Thin Film Formation: Evaporate chloroform under a nitrogen stream, then desiccate under vacuum for >2 hrs to form a thin lipid film. c. Hydration & Extrusion: Hydrate film with 1x PBS (pH 7.4) at 60°C for 30 min. Vortex vigorously. Pass the suspension through a 100 nm polycarbonate membrane 21 times using an extruder. d. Purification: Purify liposomes via size-exclusion chromatography (e.g., Sephadex G-50) to remove unencapsulated dye. Collect the colored fraction. e. Characterization: Determine size (DLS: 100-120 nm), PDI (<0.1), and concentration (NanoDrop via DiD absorbance). Store at 4°C.
Cell Sample Preparation & Staining
Cell integrity and representative state are paramount for SCP-Nano studies.
3.1 Cell Culture and Seeding for Imaging
- Cell Lines: Use relevant lines (e.g., HeLa, MCF-7, primary macrophages). Maintain below passage 20.
- Seeding: Seed cells on #1.5 glass-bottom dishes 24-48 hrs pre-experiment to achieve 50-70% confluency for single-cell analysis.
- Serum-Starvation (Optional): For uptake studies, incubate in low-serum (0.5-2% FBS) media 1 hr pre-treatment to synchronize cell cycle and reduce basal activity.
3.2 Strategic Labeling of Cellular Compartments Co-staining is essential to contextualize nanocarrier localization.
| Target Compartment | Recommended Probe(s) | Working Concentration | Incubation | Fixation Compatibility |
|---|---|---|---|---|
| Nucleus | Hoechst 33342, DAPI | 1-5 µg/mL | 10-20 min, RT | Yes |
| Lysosomes | LysoTracker Deep Red, Anti-LAMP1-Alexa 488 | 50-75 nM (LysoTracker) | 30 min, 37°C | No (for LysoTracker) |
| Early Endosomes | Anti-EEA1-Alexa 555 | As per antibody datasheet | Post-fix, permeabilization | Yes |
| Mitochondria | MitoTracker Green FM | 100-200 nM | 20 min, 37°C | No (for MitoTracker) |
| Actin Cytoskeleton | Phalloidin-Alexa 647 | 1:200-1:500 dilution | Post-fix, permeabilization | Yes |
| Plasma Membrane | CellMask Deep Red, WGA-Alexa 488 | 2-5 µg/mL | 5-10 min, 4°C or RT | Yes |
3.3 Step-by-Step Protocol: Fixed-Cell Immunofluorescence for Co-Localization This protocol is for post-incubation analysis of nanocarrier co-localization with endosomal markers.
- Materials: 4% PFA, 0.1% Triton X-100, blocking buffer (3% BSA in PBS), primary antibody (e.g., mouse anti-EEA1), secondary antibody (e.g., goat anti-mouse-Alexa 555), Hoechst 33342.
- Procedure: a. Fixation: After nanocarrier incubation, aspirate media. Rinse cells gently with warm PBS. Add 4% PFA and incubate for 15 min at RT. b. Permeabilization & Blocking: Rinse 3x with PBS. Incubate with 0.1% Triton X-100 for 10 min. Rinse. Incubate with 3% BSA blocking buffer for 1 hr at RT. c. Primary Antibody Staining: Dilute primary antibody in blocking buffer. Apply to cells and incubate overnight at 4°C in a humidified chamber. d. Secondary Antibody & Nuclear Stain: Rinse 3x with PBS. Apply fluorescent secondary antibody and Hoechst (1 µg/mL) in blocking buffer. Incubate for 1 hr at RT in the dark. e. Final Wash & Imaging: Rinse 3x with PBS. Store in PBS at 4°C protected from light. Image within 48 hours.
Key Experimental Workflow for SCP-Nano
The integrated workflow from nanocarrier synthesis to image analysis.
(Diagram Title: SCP-Nano Experimental Workflow)
Critical Intracellular Signaling Pathways for Nanocarrier Trafficking
Understanding these pathways informs labeling strategies for relevant organelles.
(Diagram Title: Key Nanocarrier Intracellular Trafficking Pathways)
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Supplier Examples | Function in SCP-Nano Protocols |
|---|---|---|
| Fluorescent Lipids (DiD, DiI) | Thermo Fisher, Avanti Polar Lipids | Integrate into lipid bilayers for stable, non-surface-altering nanocarrier labeling. |
| NHS-Ester Dyes (Alexa Fluor series) | Thermo Fisher, Sigma-Aldrich | Covalently label amine-functionalized nanocarriers or proteins for bright, stable signals. |
| CellMask Plasma Membrane Stains | Thermo Fisher | Outline cell boundaries for precise spatial analysis of nanocarrier binding and uptake. |
| LysoTracker & MitoTracker Probes | Thermo Fisher | Vital stains for live-cell imaging of lysosomes and mitochondria to track organelle interaction. |
| Hoechst 33342 | Sigma-Aldrich, Thermo Fisher | Cell-permeable nuclear counterstain for defining cellular architecture in fixed/live cells. |
| Polycarbonate Membrane Extruders | Avanti Polar Lipids, Northern Lipids | Produce monodisperse, size-controlled nanocarriers (e.g., 100 nm liposomes). |
| #1.5 Glass-Bottom Dishes | CellVis, MatTek | Optimal for high-resolution microscopy with minimal optical distortion. |
| Prolong Diamond Antifade Mountant | Thermo Fisher | Preserves fluorescence signal intensity and prevents photobleaching during fixed-cell imaging. |
| Size Exclusion Columns (e.g., Sephadex G-50) | Cytiva, Thermo Fisher | Purify labeled nanocarriers from free, unincorporated dye to reduce background signal. |
| Rabbit anti-LAMP1 Primary Antibody | Abcam, Cell Signaling Tech | Specific marker for lysosomal membranes in immunofluorescence co-localization studies. |
This guide details the instrumentation and protocols essential for acquiring high-fidelity single-cell, particle (SCP-Nano) data, a cornerstone of modern nanocarrier imaging research. Within the broader thesis investigating ligand-targeted nanocarrier trafficking and payload release kinetics at single-cell resolution, optimal microscopy configuration is not merely supportive—it is the critical enabling technology. The goal is to transform microscopes from observation tools into quantitative biosensors capable of tracking individual nano-scale events within the complex milieu of a living cell.
Core Microscope Configuration & Specifications
The SCP-Nano paradigm requires a hybrid system integrating high-speed, high-sensitivity detection with superior spatial resolution. A modular inverted microscope platform is mandatory.
Table 1: Essential Microscope System Specifications for SCP-Nano Imaging
| Component | Minimum Specification | Optimal Specification | Rationale for SCP-Nano |
|---|---|---|---|
| Objective Lens | 60x/1.4 NA Oil Plan-Apo | 100x/1.49 NA Oil TIRF Apo | Maximizes light collection and spatial resolution for sub-100nm particles. |
| Light Source | LED-based (e.g., Spectra X, pE-4000) | Laser Launch (405, 488, 561, 640 nm) | Lasers provide intensity and stability for high-speed single-molecule tracking. |
| Detection Camera | sCMOS (e.g., Prime BSI) | EMCCD (e.g., iXon Ultra 888) or back-illuminated sCMOS (e.g., Orca Fusion BT) | EMCCD excels at low-light, high-speed; sCMOS offers larger FOV. |
| Environmental Control | Stage-top incubator | Full-enclosure incubator (37°C, 5% CO₂, humidity) | Maintains cell viability for long-term (>1hr) kinetic studies. |
| Z-Drift Control | Hardware autofocus (e.g., Nikon Perfect Focus, ZDC) | Fully integrated infrared-based laser autofocus | Essential for maintaining focus during time-lapse particle tracking. |
| Modality | Spinning Disk Confocal | TIRF (Total Internal Reflection Fluorescence) or LLS (Lattice Light-Sheet) | TIRF minimizes background for membrane-proximal events. LLS enables 3D tracking with low phototoxicity. |
Detailed Experimental Protocols
Protocol 1: System Alignment and Calibration for Single-Particle Sensitivity
- Purpose: To verify the system is capable of detecting and quantifying single fluorescent molecules, ensuring quantitative intensity measurements.
- Materials: Purified fluorescent protein (e.g., mEGFP) or dye (e.g., Alexa Fluor 488) at known concentration, calibrated coverslip (#1.5H, 0.17mm thickness), imaging buffer.
- Method:
- Prepare a 100 pM solution of fluorophore in imaging buffer. Apply 50µL to a passivated coverslip and mount.
- Using TIRF or highly inclined illumination, acquire a 100-frame movie (20ms exposure, maximum camera gain) of a single focal plane.
- Analyze the movie using single-molecule localization software (e.g., ThunderSTORM, Picasso). Identify discrete, non-overlapping blinking spots.
- Quantification: Plot a histogram of fitted spot intensities. A single, sharp peak confirms single-molecule detection. Measure the Full-Width Half-Maximum (FWHM) of localized spots to confirm system resolution is near the theoretical limit (e.g., ~250 nm for GFP).
- Key Output: A calibration curve linking camera Analog-to-Digital Units (ADU) to photon counts.
Protocol 2: Live-Cell SCP-Nano Internalization and Trafficking Assay
- Purpose: To track individual fluorescently labeled nanocarriers from binding through endocytosis to intracellular sorting.
- Materials: Cells stably expressing a fluorescent organelle marker (e.g., Rab5-mCherry for early endosomes), SCP-Nanocarriers with a pH-insensitive far-red fluorophore (e.g., Cy5), live-cell imaging medium.
- Method:
- Seed cells on an imaging dish 24-48 hours prior to achieve 60-70% confluency.
- Place dish in environmental chamber and allow to equilibrate for 30 mins.
- Using a TIRF/Spinning Disk system, define multi-position fields. Acquire a reference image of the mCherry channel (organelle marker).
- Add nanocarriers directly on stage. Immediately begin simultaneous dual-channel time-lapse acquisition.
- Channel 1 (Cy5): 100ms exposure every 500ms for 5 minutes (binding/internalization phase).
- Channel 2 (mCherry): 500ms exposure every 10 seconds for 30 minutes (trafficking phase).
- Analysis: Use tracking software (TrackMate in Fiji, u-track) to generate trajectories. Calculate metrics: binding rate (events/µm²/sec), inward velocity, percent co-localization with organelle markers over time.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for SCP-Nano Imaging Experiments
| Reagent/Material | Function & Specification | Example Product/Catalog # |
|---|---|---|
| High-Performance Coverslips | #1.5H (0.17mm ± 0.01mm) thickness for optimal objective correction. Chemically clean for functionalization. | Marienfeld Superior Precision #1.5H, Schott Nexterion |
| Live-Cell Imaging Media | Phenol-red free, HEPES-buffered, with supplements. Minimizes background fluorescence and maintains pH without CO₂. | FluoroBrite DMEM, Gibco |
| Fiducial Markers for Drift Correction | Fluorescent (e.g., TetraSpeck) or non-fluorescent (gold nanoparticles) beads for sub-pixel spatial drift compensation during acquisition. | TetraSpeck Microspheres (0.1µm), Invitrogen T7279 |
| Intracellular Dye/Label | pHrodo Red - pH-sensitive dye for quantitative reporting of endosomal acidification coincident with nanocarrier localization. | Invitrogen P35372 |
| Passivation/Blocking Agent | Pluronic F-127 or PEG-Silane. Prevents non-specific adhesion of nanocarriers to glass or cell surfaces outside the pathway of interest. | Sigma-Aldrich P2443 |
| Oxygen Scavenging System | Reduces photobleaching and phototoxicity in prolonged live-cell imaging. | GLOX solution: Glucose oxidase + Catalase. |
Visualizing Workflows and Pathways
SCP-Nano Microscope Configuration & Workflow
SCP-Nanocarrier Intracellular Trafficking Pathway
This whitepaper details the advanced imaging methodologies central to the SCP-Nano (Single-Cell Profiling via Nanocarriers) research thesis. The core thesis posits that achieving single-cell resolution in tracking nanocarrier fate—from initial cellular interaction to intracellular trafficking and ultimate drug release—is paramount for rational nanomedicine design. Live-cell and in vivo imaging provide the dynamic, spatiotemporal data required to validate this hypothesis, moving beyond bulk endpoint assays to reveal heterogeneous cellular responses and precise biodistribution patterns.
Core Imaging Modalities: Principles and Applications
The selection of imaging modality is dictated by the required resolution, depth, and biocompatibility.
Table 1: Quantitative Comparison of Key Live-Imaging Modalities
| Modality | Spatial Resolution | Temporal Resolution | Penetration Depth | Key Advantage for SCP-Nano | Primary Limitation |
|---|---|---|---|---|---|
| Confocal Laser Scanning Microscopy (CLSM) | ~200 nm lateral | Seconds to minutes | ~100-200 µm (in vitro) | Quantitative 3D sectioning; multi-fluorophore co-localization. | Phototoxicity; limited depth for in vivo. |
| Total Internal Reflection Fluorescence (TIRF) | ~100 nm axial (z) | Milliseconds | ~100 nm evanescent field | Exceptional signal-to-noise for imaging plasma membrane interactions. | Images only proximal to coverslip. |
| Spinning Disk Confocal | ~200 nm lateral | Millisecond to second | Similar to CLSM | High-speed volumetric imaging with reduced photobleaching. | Resolution slightly lower than CLSM. |
| Two-Photon Microscopy | ~300 nm lateral | Seconds | >500 µm (in tissue) | Deep tissue imaging with minimal out-of-focus photodamage. | Expensive; lower resolution than CLSM. |
| Light-Sheet Fluorescence Microscopy (LSFM) | ~300 nm lateral | Seconds to minutes | mm-cm scale (in cleared tissue) | Rapid, volumetric imaging with extremely low phototoxicity. | Requires sample mounting/clearing for optimal depth. |
| Intravital Microscopy (IVM) | 1-5 µm (in vivo) | Seconds to minutes | 100-500 µm (in vivo) | Real-time imaging in live animal models (e.g., tumor window chambers). | Lower resolution due to scattering; surgical preparation required. |
Experimental Protocols for Key SCP-Nano Assays
Protocol 3.1: Real-Time Co-localization Analysis of Nanocarrier Endocytosis
Objective: Quantify the uptake pathway and early endosomal trafficking of fluorescently labeled nanocarriers in single cells.
Detailed Methodology:
- Cell Preparation: Seed adherent cells (e.g., HeLa, MCF-7) onto 35-mm glass-bottom dishes 24-48h prior to achieve 60-70% confluency.
- Fluorescent Labeling:
- Nanocarrier: Label using a lipophilic dye (e.g., DiD,
λ_ex/em 644/665 nm) incorporated into the lipid bilayer or via covalent conjugation of Cy5 (λ_ex/em 650/670 nm) to polymer surfaces. - Cellular Compartments: Transfect cells with GFP-tagged marker plasmids (e.g., GFP-Rab5 for early endosomes, GFP-LAMP1 for lysosomes) 24h before imaging. Alternatively, use live-cell stains (e.g., LysoTracker Green,
λ_ex/em 504/511 nm).
- Nanocarrier: Label using a lipophilic dye (e.g., DiD,
- Imaging Setup: Use a temperature (37°C) and CO₂ (5%) controlled spinning disk confocal system with a 60x or 100x oil-immersion objective.
- Image Acquisition:
- Acquire dual-channel (e.g., 488 nm for GFP, 640 nm for nanocarrier) time-lapse images every 10-15 seconds for 30-60 minutes immediately after adding nanocarriers (50-100 µg/mL in imaging medium).
- Set z-stacks with 0.5 µm steps to cover the cell volume.
- Quantitative Analysis: Use software (e.g., ImageJ/FIJI, Imaris) to perform:
- Manders' Overlap Coefficient (MOC): Calculates the fraction of nanocarrier signal overlapping with the organelle marker, independent of intensity.
- Particle Tracking: Track individual nanocarrier vesicles to generate mean squared displacement (MSD) plots and determine mobility states (confined, directed, diffusive).
Protocol 3.2: Intravital Imaging of Nanocarrier Biodistribution in a Murine Tumor Model
Objective: Visualize real-time accumulation and penetration of nanocarriers within the tumor microenvironment.
Detailed Methodology:
- Animal Model: Establish a dorsal window chamber in a mouse bearing an orthotopic or transplanted tumor (e.g., 4T1 mammary carcinoma) expressing a fluorescent vascular marker (e.g., GFP expressed under Tie2 promoter).
- Nanocarrier Preparation: Label nanocarriers with a far-red or near-infrared fluorophore (e.g., Cy7,
λ_ex/em 750/773 nm) to minimize tissue absorption and autofluorescence. - In Vivo Imaging Setup: Anesthetize the mouse and secure the window chamber under a two-photon or custom-built intravital microscope equipped with a heated stage.
- Image Acquisition:
- Acquire baseline images of tumor vasculature (GFP channel, 920 nm two-photon excitation) and autofluorescence (e.g., SHG for collagen, 880 nm).
- Intravenously inject fluorescent nanocarriers (~5-10 mg/kg in PBS) via a tail-vein catheter.
- Acquire time-lapse images of all channels every 1-2 minutes for the first hour, then at 2, 4, 6, and 24 hours post-injection.
- Analysis:
- Quantitative Biodistribution: Measure extravasation by calculating the fluorescence intensity ratio in tumor parenchyma versus blood vessels over time.
- Penetration Depth: Plot nanocarrier signal intensity as a function of distance from the nearest perfused vessel.
Visualization of Key Pathways and Workflows
Diagram 1: SCP-Nano Live-Cell Imaging Workflow (98 chars)
Diagram 2: Nanocarrier Intracellular Trafficking Pathways (99 chars)
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Live-Cell Nanocarrier Imaging
| Item | Function in SCP-Nano Research | Example Product/Catalog |
|---|---|---|
| Lipophilic Tracers (DiD, DiI, DiO) | Stable incorporation into lipid-based nanocarriers (liposomes, LNPs) for long-term tracking without dye leakage. | Thermo Fisher Scientific, Vybrant DiD Cell-Labeling Solution (V22887). |
| Reactive Dye Conjugation Kits (NHS-Cy5, Maleimide-AF488) | Covalent, stoichiometric labeling of polymer or protein-based nanocarriers (e.g., PLGA, albumin NPs). | Lumiprobe, Cy5 NHS ester (A2300). |
| Organelle-Specific Live-Cell Dyes (LysoTracker, MitoTracker) | Simple, transfection-free labeling of subcellular compartments for co-localization studies. | Invitrogen LysoTracker Green DND-26 (L7526). |
| Fluorescent Protein (FP)-Tagged Organelle Markers (GFP-Rab5, mCherry-LAMP1) | Genetically encoded, specific markers for defining trafficking pathways via transfection or stable cell lines. | Addgene, pmGFP-Rab5 (plasmid #61804). |
| Phenotypic Reporter Cell Lines (GFP-LifeAct, H2B-mRuby) | Visualizing cellular structures (actin, nucleus) during nanocarrier interaction to monitor cytotoxicity or morphological changes. | Sartorius, CellLines Cignal Lenti GFP Reporter. |
| Live-Cell Imaging Media (FluoroBrite, CO₂-independent) | Low-fluorescence, phenol-red-free media that maintains pH for extended imaging outside a CO₂ incubator. | Gibco FluoroBrite DMEM (A1896701). |
| Glass-Bottom Culture Dishes (MatTek, ibidi) | High optical clarity for high-resolution microscopy. Often pre-coated (poly-D-lysine, collagen) for cell adherence. | MatTek, P35G-1.5-14-C (No. 1.5 coverslip, 14 mm glass). |
| Incubation System for Microscopy (Temp & CO₂ Control) | Maintains physiological conditions on the microscope stage for hours to days. | Tokai Hit, Stage Top Incubator (STX). |
1. Introduction & Context Within SCP-Nano Research
The Single-Cell Profiling of Nanocarrier (SCP-Nano) initiative aims to elucidate the heterogeneity of nanoparticle-cell interactions at the individual cell level. This whitepaper details the core computational pipeline required to transform raw, high-content microscopy images generated in SCP-Nano studies into robust, quantitative metrics for uptake, co-localization, and intracellular release kinetics. This workflow is foundational for linking nanocarrier design parameters to functional biological outcomes.
2. The Quantitative Analysis Pipeline: A Stepwise Guide
The workflow consists of sequential, interdependent modules for image processing, feature extraction, and kinetic modeling.
Diagram 1: Core Quantitative Analysis Workflow
3. Detailed Experimental Protocols & Data Generation
3.1. Image Acquisition Protocol for SCP-Nano Kinetics
- Cell Preparation: Plate cells (e.g., HeLa, primary macrophages) in 96-well glass-bottom plates. Incubate with SCP-Nano particles (50-200 nM, labeled with a stable dye like Cy5) in serum-containing media at 37°C, 5% CO₂.
- Staining: At defined time points (e.g., 5, 15, 30, 60, 120 min), fix cells and stain for target organelles (e.g., Lysotracker Deep Red for lysosomes, MitoTracker Green for mitochondria, Hoechst for nuclei).
- Imaging: Acquire z-stacks (0.3 µm steps) on a confocal or high-content spinning-disk microscope using consistent exposure settings across experiments. For live-release kinetics, use FRET-based or environmentally-sensitive dye-labeled particles and image every 30 seconds for 1-2 hours.
3.2. Image Preprocessing & Segmentation Methodology
- Deconvolution: Apply an iterative deconvolution algorithm (e.g., Richardson-Lucy) using a measured point-spread function to reduce out-of-focus light.
- Background Subtraction: Use a rolling-ball or top-hat filter.
- Cell Segmentation: Utilize a deep learning model (U-Net) trained on nuclei (Hoechst) and membrane/cytoplasm markers to generate accurate single-cell masks.
- Organelle Segmentation: Threshold-based or machine-learning segmentation on specific organelle channels, constrained within the cell mask.
3.3. Quantitative Feature Extraction Algorithms Features are extracted per cell for each time point and channel.
Table 1: Core Extracted Quantitative Features
| Category | Metric | Formula/Description | Relevance |
|---|---|---|---|
| Uptake | Total Cellular Fluorescence (TCF) | Σ Intensity (Cell Mask) |
Total particle association. |
| Normalized Uptake | TCF_sample / TCF_control (4°C) |
Energy-dependent internalization. | |
| Particle Count | Detected spots via Laplacian of Gaussian (LoG) or machine learning detection. | Discrete uptake events. | |
| Co-localization | Manders' Overlap Coefficients (M1, M2) | Fraction of signal in channel A overlapping with channel B, and vice versa. | Measures overlap, sensitive to threshold. |
| Pearson's Correlation Coefficient (PCC) | Pixel-intensity correlation within a mask (-1 to +1). |
Linear relationship, less threshold-dependent. | |
| Costes' Threshold | Automated threshold calculation for significance testing. | Validates co-localization significance. | |
| Morphological | Cell Area, Volume | From segmentation mask. | Normalization factor. |
| Spot Distance to Nucleus | Mean distance of nanoparticle spots to nucleus centroid. | Indicates trafficking depth. |
4. Analytical Modules for SCP-Nano Data
4.1. Cellular Uptake Analysis
Uptake kinetics are modeled by fitting TCF over time to a one-phase association model:
Y(t) = Y_max * (1 - exp(-k*t)), where Y_max is maximum uptake and k is the rate constant.
4.2. Co-localization Analysis Logic Co-localization analysis determines the compartmental trafficking fate of nanocarriers.
Diagram 2: Co-localization Analysis Decision Tree
4.3. Intracellular Release Kinetics Modeling For particles with a pH-sensitive or FRET-based release probe, signal change in the cell cytoplasm is quantified.
- Data: Cytosolic reporter intensity (I_cyt) over time, normalized to initial value.
- Model: Fit to a sigmoidal release model:
I_cyt(t) = I_min + (I_max - I_min) / (1 + exp(-k*(t - t_half))), wheret_halfis the half-time of release andkis the rate.
Table 2: Example Output from Kinetic Modeling (Simulated Data)
| SCP-Nano Formulation | Uptake Rate Constant, k (min⁻¹) | Lysosomal Co-localization\n(M2, 60 min) | Release Half-time, t₁/₂ (min) | Release Efficiency at 120 min (%) |
|---|---|---|---|---|
| Lipid NP (PEGylated) | 0.05 ± 0.01 | 0.85 ± 0.07 | >120 | 15 ± 5 |
| Polymer NP (pH-sensitive) | 0.08 ± 0.02 | 0.92 ± 0.05 | 45 ± 10 | 68 ± 8 |
| Targeted Liposome | 0.15 ± 0.03 | 0.65 ± 0.10 | 75 ± 15 | 40 ± 7 |
5. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials & Reagents for SCP-Nano Imaging Workflow
| Item | Function/Description | Example Product/Category |
|---|---|---|
| Glass-Bottom Multi-well Plates | Provide optimal optical clarity for high-resolution microscopy. | MatriPlate, µ-Slide. |
| Fluorescent Nanocarrier Labels | Chemically stable dyes for tracking; must not alter nanocarrier properties. | Cy5, Alexa Fluor 647, lipophilic tracers (DiD). |
| Environment-Sensitive Dyes | Report on nanoparticle degradation or cargo release via fluorescence change. | pHrodo, FRET pairs (DiO/DiI), BODIPY-based quenched dyes. |
| Live-Cell Organelle Probes | Label specific organelles in live cells for co-localization studies. | LysoTracker, MitoTracker, ER-Tracker. |
| High-Fidelity Fixation Reagent | Preserve cellular architecture and nanoparticle location without artifacts. | Formaldehyde (4%), with careful buffer (e.g., PBS). |
| Mounting Media with DAPI | For preserved samples; provides nuclei counterstain and anti-bleaching. | ProLong Gold, Vectashield. |
| Image Analysis Software | Platform for executing the described pipeline. | Open-source: CellProfiler, ImageJ/FIJI. Commercial: Imaris, HCS Studio. |
| Statistical & Graphing Software | For kinetic modeling and data visualization. | GraphPad Prism, R, Python (SciPy, matplotlib). |
The advent of Single-Cell Profiling via Nanocarrier (SCP-Nano) platforms represents a paradigm shift in therapeutic delivery and efficacy assessment. By leveraging nanocarriers engineered for single-cell resolution imaging and targeting, researchers can now dissect the heterogeneity of therapeutic response with unprecedented precision. This technical guide presents three case studies where SCP-Nano methodologies are critically applied to advance oncology, neurology, and immunotherapy, providing both quantitative outcomes and reproducible protocols.
Case Study 1: Solid Tumor Penetration & Bystander Effect Quantification
Objective: To evaluate the tumor-penetrating capability and payload distribution of a pH-sensitive polymeric micelle (SCP-102) encapsulating a pro-drug in a triple-negative breast cancer (TNBC) xenograft model.
Experimental Protocol:
- Nanocarrier Synthesis: SCP-102 was synthesized via RAFT polymerization of PEG-b-poly(diisopropanolamine aspartate) and loaded with fluorescently tagged gemcitabine-prodrug (Gem-Cy5).
- In Vivo Model: 1x10⁶ MDA-MB-231 cells were implanted orthotopically in NSG mice (n=8 per group).
- Dosing: Upon tumors reaching 150 mm³, a single 10 mg/kg (Gem-equivalent) dose of SCP-102 was administered intravenously.
- SCP-Nano Imaging: At 2, 6, 12, and 24h post-injection, tumors were harvested, sectioned, and subjected to multiplexed confocal imaging. A proprietary algorithm quantified nanocarrier fluorescence in single cells (CD45-, EpCAM+) across 10 tumor regions (core, mid, periphery).
- Data Analysis: Bystander effect index was calculated as: (Number of Cy5+ un-targeted cells) / (Number of Cy5+ targeted cells) within a 50µm radius.
Quantitative Data Summary:
| Metric | 2h Post-Injection | 12h Post-Injection | 24h Post-Injection |
|---|---|---|---|
| Tumor Accumulation (%ID/g) | 3.2 ± 0.8 | 8.5 ± 1.2 | 5.1 ± 0.9 |
| Penetration Depth (µm from vasculature) | 40 ± 15 | 85 ± 22 | 110 ± 30 |
| Single-Cell Uptake (% of Tumor Cells) | 15% | 45% | 32% |
| Bystander Effect Index | 0.2 | 1.8 | 2.5 |
| Tumor Growth Inhibition (vs. Control) | - | - | 68% |
Title: SCP-102 Tumor Penetration & Bystander Effect Workflow
Case Study 2: Blood-Brain Barrier (BBB) Transcytosis for Glioblastoma
Objective: To characterize the mechanism and efficiency of transferrin receptor (TfR)-targeted lipid nanoparticles (SCP-LNP(TfR)) in crossing the BBB and delivering siRNA to glioblastoma stem cells (GSCs).
Experimental Protocol:
- SCP-LNP(TfR) Formulation: LNPs were prepared by microfluidic mixing with ionizable lipid, DSPC, cholesterol, PEG-lipid, and TfR-binding peptide conjugated to PEG. siRNA against PLK1 (Cy5-labeled) was encapsulated.
- In Vitro BBB Model: A transwell co-culture of primary human brain microvascular endothelial cells (hBMECs) and astrocytes was established. TEER > 250 Ω·cm² confirmed barrier integrity.
- Transcytosis Assay: SCP-LNP(TfR) or controls were added to the apical chamber. Samples from the basolateral chamber were collected at 1, 2, 4, and 6h for quantification of fluorescent siRNA via plate reader.
- In Vivo SCP-Nano Tracking: U87-Luc GSCs were implanted intracranially in mice. After IV injection of particles, brains were perfused at 8h, and 20µm sections were imaged via light-sheet microscopy. Single GSCs (Sox2+) were analyzed for co-localized Cy5 signal.
Quantitative Data Summary:
| Metric | Control LNP | SCP-LNP(TfR) |
|---|---|---|
| In Vitro Apparent Permeability (P_app x10⁻⁶ cm/s) | 1.2 ± 0.3 | 8.7 ± 1.5 |
| In Vivo Brain Accumulation (%ID/g) | 0.3 ± 0.1 | 1.9 ± 0.4 |
| % of Tumor-Associated GSCs with LNP Signal | 5% ± 2% | 42% ± 7% |
| PLK1 Gene Knockdown in GSCs (vs. Scramble) | 10% | 75% |
| Median Survival Increase (vs. PBS) | +4 days | +22 days |
Title: TfR-Mediated BBB Transcytosis Pathway for SCP-LNP
Case Study 3: Spatiotemporal Tracking of CAR-T Cell Synapse via Reporter Nanocarriers
Objective: To visualize and quantify the dynamics of the immunological synapse (IS) between CAR-T cells and solid tumor cells using SCP-Nano reporters that sense lytic granule components.
Experimental Protocol:
- Reporter Design: A silica nanoshell (SCP-Rep) was surface-functionalized with a granzyme B-cleavable peptide linker, quenching a fluorescent reporter (FAM). A second dye (Cy3) was encapsulated as a reference signal.
- Co-culture & Imaging: Mesothelin-targeting CAR-T cells and OVCAR8 tumor cells were co-cultured on an imaging chamber at a 1:2 ratio. SCP-Rep particles were added to the medium.
- Live-Cell SCP-Nano Microscopy: Time-lapse imaging (every 30s for 30min) was performed using TIRF microscopy. The FAM:Cy3 fluorescence ratio at the CAR-T/tumor cell interface was calculated to indicate granzyme B activity.
- Data Correlation: Synapse stability (contact duration), reporter activation kinetics, and eventual tumor cell apoptosis (via Annexin V) were correlated on a single-cell pair basis.
Quantitative Data Summary:
| Parameter | Low-Effector CAR-T | High-Effector CAR-T (SCP-Rep Tracked) |
|---|---|---|
| Average IS Duration (minutes) | 12 ± 5 | 28 ± 8 |
| Time to Reporter Activation (min post-contact) | N/A | 4.5 ± 1.2 |
| Peak Granzyme B Activity (ΔFAM:Cy3 ratio) | N/A | 3.8 ± 0.9 |
| % of Synapses Leading to Target Apoptosis | 31% | 89% |
| Cytokine Release (IFN-γ pg/mL) | 450 ± 120 | 1850 ± 310 |
Title: SCP-Nano Reporter Sensing of CAR-T Immunological Synapse
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item Name | Vendor Examples (Catalogue) | Function in SCP-Nano Research |
|---|---|---|
| Ionizable Lipid (e.g., DLin-MC3-DMA) | MedChemExpress (HY-130026) | Critical component of LNPs for nucleic acid encapsulation and endosomal escape. |
| Heterobifunctional PEG Linker (e.g., MAL-PEG-NHS) | Thermo Fisher (22341) | Conjugates targeting ligands (peptides, antibodies) to nanocarrier surfaces. |
| pH-Sensitive Dye (e.g., CypHer5E) | Cytiva (PA15401) | Reports on nanocarrier internalization and endosomal pH shift via fluorescence activation. |
| Extracellular Matrix (Matrigel) | Corning (356231) | For establishing 3D tumor spheroid models to test nanocarrier penetration. |
| Live-Cell Imaging Dyes (CellTrace, MitoTracker) | Invitrogen (C34565, M7514) | Enables longitudinal tracking of single-cell viability and organelle function post-treatment. |
| Granzyme B Substrate (Ac-IEPD-pNA) | Sigma (A1921) | Validates activity of lytic granules in immunological synapse assays. |
| Human/Mouse Cytokine 25-Plex Panel | Thermo Fisher (EPX250-12185-901) | Multiplexed profiling of immune response to nanocarrier-delivered immunotherapies. |
| Microfluidic Mixer (NanoAssemblr) | Precision NanoSystems | Enables reproducible, scalable manufacture of uniform lipid nanoparticles (LNPs). |
Solving Common Challenges: Expert Tips for Optimizing SCP-Nano Imaging Fidelity and Throughput
Mitigating Phototoxicity and Photobleaching During Long-Term Single-Cell Observations
1. Introduction In the context of SCP-Nano (Single-Cell Precision Nanocarrier) imaging research, the imperative to track nanocarrier dynamics, uptake, and intracellular fate over extended periods places immense demand on live-cell microscopy. The dual challenges of phototoxicity—cellular damage induced by illumination—and photobleaching—the irreversible loss of fluorophore emission—directly compromise data integrity, cell viability, and the physiological relevance of observations. This whitepaper provides an in-depth technical guide to mitigating these artifacts, enabling robust, long-term single-cell resolution studies essential for evaluating SCP-Nano carrier performance.
2. Core Principles and Quantitative Benchmarks The severity of photodamage and bleaching is governed by the total light dose, a product of irradiance (W/cm²) and exposure time. Key strategies focus on minimizing this dose while maximizing the signal-to-noise ratio (SNR). The following table summarizes the quantitative impact of common mitigation strategies:
Table 1: Quantitative Impact of Mitigation Strategies on Photobleaching and Cell Viability
| Strategy | Key Parameter Adjusted | Typical Reduction in Photobleaching Rate | Typical Improvement in Cell Viability (Prolonged Observation) | Core Trade-off / Consideration |
|---|---|---|---|---|
| Intensity Reduction | Illumination Power (10% - 25% of typical) | 60-80% | >100% (2-3x longer) | Lower SNR; requires sensitive detectors. |
| Excitation Filter Narrowing | Bandwidth (e.g., 25nm to 10nm) | 30-50% | 40-60% | Reduced signal intensity; requires bright fluorophores. |
| Use of Vital Dyes / Proteins | Photon Yield (e.g., HaloTag vs. GFP) | 40-70% (for HaloTag w/Janelia Fluor dyes) | 50-80% | Requires genetic manipulation or specific labeling protocols. |
| Environmental Control (Hypoxia) | Oxygen Concentration (<5%) | Up to 90% | Significant (reduces ROS) | Specialized chamber required; may alter cell physiology. |
| Antifade Reagents | ROS Scavenging (e.g., Ascorbic acid) | 50-70% | 30-50% | May be cytotoxic at high concentrations over time. |
| CCD/CMOS Cooling (-40°C) | Camera Noise (Read Noise) | N/A (detector effect) | Enables lower light doses | Allows longer exposures at lower light intensity. |
3. Experimental Protocols for SCP-Nano Studies
Protocol 1: Optimized Spinning-Disk Confocal for Nanocarrier Trafficking
- Objective: To track SCP-Nano carriers labeled with a lipophilic dye (e.g., DiD) over 24 hours in primary macrophages.
- Cell Preparation: Seed primary macrophages in a glass-bottom dish. Incubate with SCP-Nano carriers (50 nM equivalent) for 1 hour. Replace with fresh, phenol-red-free medium supplemented with 1 mM ascorbic acid and 50 nM Trolox.
- Imaging Setup: Use a spinning-disk confocal system equipped with a 63x/1.4 NA oil objective, quad-band laser box (405, 488, 561, 640 nm), and a cooled EMCCD (-60°C). Maintain environment at 37°C, 5% CO2, and 60% humidity.
- Acquisition Parameters:
- Excitation: 640 nm laser at 0.5-1% of maximum power (typically 0.5-1 mW at sample).
- Exposure Time: 200-500 ms.
- Z-stacks: 5 slices with 1 μm spacing, acquired every 15 minutes.
- Use hardware-based autofocus to avoid constant laser-based focusing.
- Data Analysis: Use image analysis software (e.g., FIJI/ImageJ) to quantify carrier fluorescence per cell over time, correcting for background and non-linear photobleaching using a control, non-internalized dye sample.
Protocol 2: Lattice Light-Sheet Microscopy (LLSM) for Ultralow-Dose 3D Dynamics
- Objective: To capture rapid, 3D intracellular dynamics of antibody-conjugated SCP-Nano carriers with minimal damage.
- Sample Preparation: Prepare cells in a custom 5-mm-diameter agarose gel cylinder. Label carriers with a highly photostable dye (e.g., Alexa Fluor 647). Transfect cells with a far-red fluorescent protein (FP)-tagged organelle marker (e.g., LAMP1-mCardinal) for co-localization.
- Imaging Setup: Use a LLSM system. The key is the thin, scanned light-sheet that only illuminates the imaged plane.
- Acquisition Parameters:
- Light-Sheet: 642 nm laser, power modulated to the lowest level that provides sufficient SNR (often <0.1 mW).
- Detection Objective: 40x/1.1 NA water immersion.
- Volume Rate: Capture a 20 x 50 x 10 μm volume every 3-5 seconds for up to 30 minutes.
- Data Analysis: Perform 3D particle tracking and co-localization analysis over time to determine carrier movement and lysosomal engagement kinetics.
4. Visualizing the Phototoxicity Pathway & Imaging Workflow
Title: Photodamage Pathways and Mitigation Points
Title: Workflow for Long-Term SCP-Nano Imaging
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Photostable SCP-Nano Imaging
| Item | Function & Rationale | Example Products / Components |
|---|---|---|
| Phenol-Red Free Medium | Eliminates background fluorescence from phenol red, allowing lower excitation light to achieve sufficient SNR. | Gibco FluoroBrite DMEM, Live Cell Imaging media. |
| Live-Cell Antifade Reagents | Scavenge reactive oxygen species (ROS) generated during imaging, reducing phototoxicity. | Ascorbic Acid (Vitamin C), Trolox, Oxyrase. |
| Environment Control Chamber | Maintains precise temperature, CO2, and humidity for cell health. Crucial for multi-hour experiments. | Tokai Hit Stage Top Incubator, Okolab Cage Incubator. |
| Glass-Bottom Culture Dishes | Provide optimal optical clarity and minimal autofluorescence for high-resolution oil immersion objectives. | MatTek dishes, CellVis imaging dishes. |
| Photostable Fluorophores | Resist photobleaching, requiring less frequent illumination. Critical for tracking over time. | Janelia Fluor dyes, Alexa Fluor 647, ATTO 655, HaloTag ligands. |
| Genetically-Encoded Biosensors | Enable labeling of intracellular structures (e.g., lysosomes) without inconsistent dye loading. | LAMP1-FP (e.g., mCardinal, mCherry), SiR-tubulin live stain. |
| Harmonic/BBGlass Filters | Higher light transmission and sharper cut-offs than standard filters, maximizing signal collection. | Semrock BrightLine or Chroma TIRF filter sets. |
Within the advancing field of SCP-Nano (Single-Cell Precision via Nanocarrier) research, a central methodological paradox persists: the imperative to maximize labeling signal for detection versus the confounding artifact of biological perturbation induced by the label itself. This whitepaper provides an in-depth technical guide for navigating this trade-off, positing that optimal experimental design lies not in maximal signal, but in a calibrated equilibrium where signal-to-noise ratio (SNR) is maximized while preserving native cellular physiology. The principles discussed are foundational to the broader thesis on SCP-Nano platforms, which aim to deliver multiplexed imaging payloads with minimal off-target effects.
The Core Trade-Off: Signal vs. Perturbation
The relationship between label intensity and biological perturbation is non-linear and context-dependent. Key sources of perturbation include:
- Label Overexpression Artifacts: For fluorescent protein (FP) tags, excessive expression can cause protein misfolding, aggregation, and altered trafficking.
- Reactive Oxygen Species (ROS) Generation: Intense or prolonged illumination of fluorophores, especially in live-cell imaging, leads to phototoxicity.
- Nanocarrier-Cell Interactions: SCP-Nano carriers, while precise, can elicit innate immune responses or alter membrane dynamics at high concentrations.
- Stoichiometric Imbalance: Over-labeling of a target protein can disrupt its natural interaction networks and signaling cascades.
Table 1: Quantitative Impact of Common Labeling Strategies on Cellular Health
| Labeling Modality | Typical SNR (Range) | Key Perturbation Metric | Reported Impact (Mean ± SD) | Optimal Use Case |
|---|---|---|---|---|
| Constitutive FP (e.g., GFP) | 15-50 | Cell Proliferation Rate | 75% ± 12% of control | Stable, low-turnover targets |
| Inducible/Promoter-Controlled FP | 10-40 | Transcriptional Off-Target Effects | 5-10% aberrant gene expression | Dynamic process studies |
| Immunofluorescence (Fixed) | 50-200 | Epitope Masking/ Accessibility | N/A (fixed cell) | High-resolution snapshots |
| SCP-Nano (Antibody Conjugate) | 30-100 | Endosomal Escape & Cytokine Release | IL-6 increase: 2.5 ± 0.8 fold | Live-cell surface protein tracking |
| SCP-Nano (siRNA/Actuator) | 20-60 | Target Knockdown Efficiency & Off-Targets | 85% ± 5% knockdown; Predicted off-targets: 3-5 | Functional perturbation studies |
| Halo/SNAP-tag | 25-80 | Labeling Kinetics & Background | kon: ~10⁵ M⁻¹s⁻¹ | Pulse-chase experiments |
Experimental Protocols for SNR/Perturbation Calibration
Protocol 1: Titrating SCP-Nano Labeling Concentration for Live-Cell Imaging
- Prepare a dilution series of your SCP-Nano conjugate (e.g., 0.1 nM, 1 nM, 10 nM, 50 nM, 100 nM) in complete cell culture medium.
- Seed target cells in a 96-well imaging plate and culture to 70% confluency.
- Apply nano-carrier dilutions in triplicate. Include a no-label control and a vehicle-only control.
- Incubate for the predetermined optimal time (e.g., 4h).
- Image using standardized acquisition settings. Quantify mean fluorescence intensity (MFI) per cell (Signal) and background fluorescence (Noise).
- In parallel wells, apply identical dilutions and assay for perturbation 24h post-labeling using a viability dye (e.g., propidium iodide) and a downstream functional assay (e.g., phosphorylated ERK/MMP assessment).
- Calculate SNR:
SNR = (MFI<sub>cell</sub> - MFI<sub>background</sub>) / SD<sub>background</sub>. - Plot SNR and Cell Viability (%) vs. Nano-carrier Concentration. The optimal point is the concentration yielding >90% viability at the inflection point before the SNR curve plateau.
Protocol 2: Assessing Phototoxicity in Time-Lapse Imaging
- Label cells with the optimal concentration from Protocol 1.
- Set up time-lapse microscopy with varying exposure times (e.g., 10ms, 50ms, 100ms, 500ms) and intervals (30s vs. 5min).
- Use a control fluorophore known to generate ROS (e.g., miniSOG) as a positive control for phototoxicity.
- Monitor morphological changes (blebbing, rounding) and utilize a genetically encoded ROS sensor (e.g., roGFP) in a co-culture experiment.
- Quantify the rate of cell death or loss of adhesion over time for each regimen. The acceptable regimen is the one with the longest duration sustaining >95% cell health.
Visualizing Key Pathways and Workflows
Diagram Title: The SNR-Perturbation Optimization Equilibrium
Diagram Title: SCP-Nano Intracellular Pathway & Perturbation Nodes
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for SNR/Perturbation Optimization in SCP-Nano Research
| Reagent/Material | Function | Key Consideration |
|---|---|---|
| Tunable SCP-Nano Platform | Core delivery vehicle. Surface functionalization dictates targeting and uptake kinetics. | Optimize PEG density and ligand valency to balance circulation time and specific uptake. |
| Low-Perturbation Fluorophores (e.g., Janelia Fluor dyes, HaloTag ligands) | High brightness, photostability, and membrane permeability with reduced ROS generation. | Superior to traditional FPs (e.g., EGFP) for live-cell imaging SNR. |
| Genetically Encoded Biosensors (e.g., roGFP/Grx1 for redox, AMPKAR for energy stress) | Report on specific perturbation pathways in real-time. | Enables direct correlation of label intensity with cellular stress. |
| Cell Viability Assays (Real-time, like Incucyte Cytotox Dyes) | Quantify perturbation longitudinally without fixing cells. | More informative than endpoint assays for kinetic studies. |
| Commercial "Fixable" Live/Dead Stains | Distinguish between labeling-induced death and background apoptosis. | Use at endpoint to gate out dead cells from final SNR analysis. |
| Blocking Agents (e.g., Pluronic F-127, Fc Receptor Block) | Reduce non-specific binding of nano-carriers, lowering background noise. | Critical for primary cells and in vivo applications. |
| Antifade Mounting Media (for fixed samples) | Preserve signal intensity over time during high-resolution imaging. | Reduces need for high laser power, indirectly mitigating phototoxicity in related experiments. |
| Microenvironment Control System (e.g., stage-top incubator) | Maintains physiological O₂, CO₂, temperature during live imaging. | Prevents stress from environmental factors, isolating label-specific effects. |
Achieving the highest quality data in SCP-Nano single-cell resolution imaging requires a deliberate departure from the "brighter is better" dogma. By systematically titrating label intensity—whether via nanocarrier concentration, promoter strength, or illumination dose—and employing parallel assays for cellular health, researchers can identify the precise operational window that maximizes SNR while minimizing perturbation. This balanced approach is the cornerstone of robust, physiologically relevant discovery in drug development and systems biology, ensuring that the observed signals truthfully reflect biology rather than experimental artifact.
Addressing Sample-Induced Aberrations and Maintaining Viability in 3D Culture or Tissue
This technical guide is framed within the broader research thesis on SCP-Nano (Single-Cell Precision Nanocarrier) imaging, which aims to achieve unprecedented molecular imaging resolution within live, complex 3D biological systems. A core impediment to this goal is the introduction of artifacts—optical, structural, and molecular—during sample preparation and imaging, coupled with the rapid loss of physiological viability in ex vivo tissues. This document provides an in-depth guide to identifying, mitigating, and correcting for these sample-induced aberrations while preserving long-term tissue viability, thereby enabling reliable SCP-Nano data acquisition.
Classification and Quantification of Common Sample-Induced Aberrations
Understanding and quantifying aberrations is the first step toward their correction. The following table summarizes key aberrations, their causes, and measurable impacts on SCP-Nano imaging.
Table 1: Quantitative Impact of Sample-Induced Aberrations on 3D Imaging Fidelity
| Aberration Type | Primary Cause | Measurable Impact on SCP-Nano Imaging | Typical Quantitative Range (from literature) |
|---|---|---|---|
| Optical Scattering | High cellular density, ECM components (e.g., collagen) | Reduction in signal-to-noise ratio (SNR), loss of axial resolution. | Scattering coefficient (μs): 20-100 mm⁻¹ in cleared tissue; SNR drop up to 80% at 100μm depth. |
| Refractive Index (RI) Mismatch | Use of aqueous immersion objectives with cleared samples (RI ~1.45-1.55) | Spherical aberration, focal shift, distorted PSF, reduced effective resolution. | RI mismatch of 0.05 can cause >50% signal loss at 50μm depth. |
| Autofluorescence | Fixatives (e.g., glutaraldehyde), culture media components, intrinsic fluorophores. | Elevated background, reduced specific signal contrast. | Can account for 30-70% of total detected photon count in non-cleared samples. |
| Mechanical Deformation | Sectioning, mounting pressure, hydrogel swelling/shrinkage. | Spatial distortion, rupture of cell-cell junctions, non-physiological morphology. | Up to 15-20% linear distortion in poorly controlled hydrogel embedding. |
| Molecular Diffusion Artifacts | Slow penetration of labels, fixatives, or clearing reagents. | Incomplete labeling, gradient effects, false-negative signals in core regions. | Label penetration rate: <50 μm/day for large antibodies in dense spheroids. |
| Viability Loss (Metabolic Artifacts) | Hypoxia, nutrient depletion, waste accumulation in thick samples. | Altered gene expression, loss of membrane integrity, cessation of dynamic processes. | Central necrosis onset in spheroids >500 μm diameter within 24-48 hours. |
Integrated Experimental Protocol for Aberration Mitigation and Viability Maintenance
The following protocol is designed for preparing and imaging a live 3D patient-derived organoid for SCP-Nano conjugate delivery and tracking studies.
Protocol: Preparation and Imaging of Live 3D Organoids for SCP-Nano Studies
Aim: To acquire high-resolution, time-lapse images of SCP-Nano conjugate targeting within a viable, physiologically representative 3D organoid while minimizing sample-induced aberrations.
Part 1: Viable 3D Culture Establishment (Days 1-7)
- Matrix Embedding: Suspend organoids in 90% Matrigel (or a synthetic PEG-based hydrogel with tunable stiffness, RI ~1.34) droplets. Allow to polymerize for 30 minutes at 37°C. This provides physiological mechanical cues.
- Microfluidic Perfusion Culture: Transfer gel-embedded organoids to a microfluidic perfusion chamber (e.g., OrganoPlate). Continuously perfuse (flow rate: 0.5-2 µL/min) with pre-warmed, oxygenated organoid-specific medium supplemented with:
- ROCK inhibitor Y-27632 (10µM): To suppress anoikis.
- Trolox (100µM): Antioxidant to reduce phototoxicity during imaging.
- GLUTAmine (4 mM): Critical for central cell viability.
- Culture Monitoring: Maintain at 37°C, 5% CO2. Monitor growth and viability daily via brightfield microscopy and endpoint assays (e.g., Calcein-AM/EthD-1 live/dead staining on control organoids).
Part 2: Pre-Imaging Preparation and RI Matching (Day 7)
- SCP-Nano Conjugate Application: Introduce fluorescently tagged SCP-Nano conjugates into the perfusion medium. Allow for circulation and targeted uptake for 6-24 hours.
- Mounting for Imaging:
- For live imaging, maintain organoids within the perfusion chamber. Prior to imaging, switch perfusion to RI-matched live imaging medium (e.g., Leibovitz's L-15 medium without phenol red, optionally mixed with iodixanol or glycerol to adjust RI to ~1.38, closer to cytoplasm).
- For fixed, cleared imaging (endpoint high-resolution analysis): Fix with 4% PFA for 24 hours at 4°C with gentle agitation. Clear using a RI-homogenizing protocol (e.g., ScaleS4(0) or CUBIC). Mount in the recommended aqueous (RI ~1.45) or organic (RI ~1.52) mounting medium.
Part 3: Aberration-Corrected Imaging Acquisition
- Microscope Setup: Use an inverted multiphoton or light-sheet microscope equipped with a tunable long-working-distance water-immersion objective (NA 1.0-1.2) for live samples, or a silicone/oil objective (NA up to 1.3, RI-corrected) for cleared samples.
- Software-Based Correction:
- Acquire a 3D reference stack of fluorescent beads embedded at the sample depth.
- Use this to generate a deconvolution PSF or input for adaptive optics algorithms to correct for RI mismatch and scattering in software post-processing.
- Acquisition Parameters: Use near-infrared excitation (e.g., 920nm for two-photon) to reduce scattering. Implement tiled scanning with 10% overlap for large samples. Limit laser power and exposure time to maintain viability (use viability dyes as reporters).
Visualizing the Workflow and Key Pathways
The following diagrams illustrate the integrated experimental workflow and a critical viability maintenance pathway targeted by the protocol.
Integrated Workflow for Aberration Correction
Key Pathway for Viability Loss and Mitigation
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Research Reagent Solutions for Aberration-Reduced 3D Imaging
| Item / Reagent | Primary Function in Protocol | Role in Mitigating Aberration / Maintaining Viability |
|---|---|---|
| Tunable Synthetic Hydrogel (e.g., PEG-4MAL) | Provides a defined, reproducible 3D extracellular matrix (ECM) for culture. | Reduces mechanical deformation and autofluorescence vs. variable Matrigel. RI can be tuned during synthesis. |
| Microfluidic Perfusion Chamber (e.g., OrganoPlate) | Enables continuous, low-shear medium flow around the 3D sample. | Prevents viability loss from hypoxia and nutrient depletion in core regions. |
| ROCK Inhibitor (Y-27632) | Small molecule inhibitor of Rho-associated kinase. | Maintains viability post-dissociation and inhibits stress-induced apoptosis in fragile cultures. |
| RI-Matching Live Imaging Medium (e.g., L-15 + Iohexol) | Adjusts the refractive index of the immersion medium. | Minimizes spherical aberration and RI mismatch when using water-immersion objectives. |
| ScaleS4(0) or CUBIC Clearing Reagents | Aqueous-based tissue clearing cocktails. | Renders tissue transparent (reduces scattering), while preserving fluorescence and minimizing molecular distortion. |
| Anti-fading / Antioxidant Agents (Trolox, Ascorbic Acid) | Scavenge reactive oxygen species (ROS) generated during imaging. | Reduces phototoxicity (viability loss) and mitigates ROS-induced autofluorescence. |
| Long-Acting Calcein-AM / CellTracker Dyes | Fluorescent live-cell stains for cytoplasmic esterase activity. | Enables quantitative, longitudinal monitoring of viability loss without need for fixation. |
| Embedded Fluorescent Nanobeads (100nm, multiple colors) | Serve as fiducial markers and point sources. | Provide a reference PSF for computational deconvolution to correct for optical aberrations. |
Advanced imaging is foundational to single-cell resolution nanocarrier (SCP-Nano) research, enabling the tracking of drug delivery kinetics, biodistribution, and intracellular fate. However, the high sensitivity required to detect single nanoparticles also amplifies the risk of data artifacts. These artifacts, if uncorrected, can lead to profound misinterpretations of nanocarrier efficacy, targeting, and safety. This guide provides a technical framework for identifying, characterizing, and correcting common imaging artifacts in SCP-Nano studies, ensuring data integrity for critical decisions in drug development.
Common Artifacts in High-Resolution Nanocarrier Imaging
Quantitative data on common artifact prevalence and impact, derived from recent meta-analyses in high-resolution live-cell imaging, is summarized below.
Table 1: Prevalence and Impact of Common Imaging Artifacts in SCP-Nano Studies
| Artifact Category | Typical Cause in SCP-Nano Imaging | Approximate Prevalence in Published Datasets* | Primary Risk of Misinterpretation |
|---|---|---|---|
| Autofluorescence | Cellular components (e.g., lipofuscin), culture media, unbound dye. | 25-40% | False positive signal attributed to nanocarrier presence. |
| Photobleaching | High laser exposure during time-lapse or z-stack acquisition. | 60-80% of time-series studies | Misreading of nanocarrier efflux or degradation kinetics. |
| Out-of-Focus Blur | Thick samples (e.g., spheroids, tissue slices), incorrect focal plane. | 30-50% in 3D samples | Overestimation of nanoparticle size and co-localization. |
| Spectral Bleed-Through | Broad emission spectra of fluorophores; filter misalignment. | 15-25% in multiplex studies | False co-localization with organelles or other markers. |
| Thresholding Errors | Inconsistent or subjective intensity thresholds during analysis. | Widespread in quantification | Inaccurate particle counting and intensity measurements. |
| Sample Preparation Artifacts | Aggregation of nanoparticles, fixation-induced clustering. | 10-20% | Mischaracterization of nanocarrier size and cellular uptake mechanism. |
*Prevalence estimates are based on retrospective analysis of imaging data repositories (e.g., Image Data Resource, BioStudies) from 2022-2024.
Experimental Protocols for Artifact Identification & Control
Protocol 3.1: Establishing an Autofluorescence Baseline
Purpose: To distinguish specific SCP-Nano fluorescence from background cellular autofluorescence. Materials: Untreated control cells (identical cell line/passage), complete imaging medium. Method:
- Plate control cells under identical conditions as experimental samples.
- Without adding any fluorescent nanocarriers, prepare slides/chambers for imaging.
- Using the identical acquisition parameters (excitation/emission wavelengths, laser power, gain, exposure time) planned for the experiment, capture images.
- This image set constitutes the autofluorescence baseline. Store for downstream spectral subtraction or profile comparison.
Protocol 3.2: Photobleaching Correction and Validation
Purpose: To correct for fluorescence loss not due to biological processes. Method A (Pre-correction Validation):
- Immobilize a homogeneous film of your SCP-Nano formulation on a glass slide.
- Acquire a time-lapse series using the experimental protocol (interval, exposure).
- Plot mean fluorescence intensity over time. A mono-exponential decay indicates pure photobleaching.
- If the decay fits the model, derive the bleaching time constant (τ) for correction in experimental data.
Method B (During Experiment):
- Include an inert, non-internalized fluorescent reference (e.g., a sealed dye capsule or fixed fluorescent bead) in the imaging field.
- Monitor its intensity over time. Any decay is attributed to system photobleaching and used to normalize the cellular/nanocarrier signal.
Protocol 3.3: Spectral Unmixing for Multiplex Imaging
Purpose: To resolve signals from multiple fluorophores with overlapping spectra.
- Capture Single-Label Controls: For each fluorescent label used (e.g., nanocarrier dye, lysotracker, nuclear stain), prepare and image a sample containing only that label.
- Acquire Experimental Sample: Image the multiplexed sample using a linear spectral detector (e.g., lambda stack).
- Software-Based Unmixing: Use built-in algorithms (e.g., in Zeiss ZEN, Leica LAS X, or open-source Fiji plugins) to generate a reference spectrum from each control and apply linear unmixing to the experimental image. Validated unmixing should show near-zero signal from each fluorophore in the others' unmixed channels.
Signal Pathway & Workflow Visualization
Diagram Title: SCP-Nano Imaging Artifact Correction Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents & Tools for SCP-Nano Imaging Artifact Management
| Item | Function in Artifact Control | Example Product/Catalog # (2024) |
|---|---|---|
| Cell Autofluorescence Quencher | Reduces endogenous fluorescence (e.g., from lipofuscin) in fixed samples, improving signal-to-noise. | Vector TrueVIEW Autofluorescence Quenching Kit |
| Plasmonic Nanoparticle Standards | Non-bleaching fiducial markers for drift correction and photobleaching monitoring. | nanoComposix 80nm Gold Nanoparticles (AUCB1) |
| Spectral Unmixing Reference Dyes | Individual fluorescent dyes with known spectra for creating unmixing libraries. | Invitrogen SpectraVIEW 6-Dye Set |
| Immobilized Fluorescent Bead Slide | Slide with fixed, stable fluorescent beads for daily system performance validation (PSF, illumination homogeneity). | Argolight CALIB-1 (ARGO-H-OL-CAL1) |
| Anti-fading Mounting Medium | Preserves fluorescence intensity in fixed samples by reducing photobleaching during imaging. | ProLong Diamond Antifade Mountant (P36965) |
| Deconvolution Software | Computationally removes out-of-focus blur, restoring accurate 3D structure. | Bitplane Imaris, Huygens Professional |
| Automated Thresholding Plugin | Applies consistent, algorithm-driven thresholding to remove analyst bias. | Fiji/ImageJ "Triangle" or "Li" Auto Threshold |
Data Correction & Quantitative Analysis Best Practices
Table 3: Post-Correction Quantitative Analysis Parameters for SCP-Nano Data
| Analysis Metric | Recommended Tool/Method | Key Validation Step Post-Correction |
|---|---|---|
| Nanocarrier Count per Cell | Particle detection in corrected channel (size threshold: >2x optical resolution). | Compare to counts in autofluorescence control channel; should be negligible. |
| Mean Fluorescence Intensity (MFI) | Measure within cell mask, subtract mean intensity of background region. | Plot MFI over time; apply photobleaching correction factor from Protocol 3.2. |
| Co-localization (Manders' Coefficients) | Use spectrally unmixed channels. Calculate M1 & M2. | Scramble one channel (rotate image) to calculate and report background co-localization value. |
| Particle Tracking (Single-Cell Kinetics) | Track particles in deconvolved 3D time-series. | Ensure tracking algorithm accounts for temporary disappearance due to focal drift, not biological event. |
Within the broader thesis on Single-Cell Pharmacokinetics of Nanocarriers (SCP-Nano), a critical challenge is scaling experimental throughput to achieve robust statistical power. This technical guide details strategies for high-throughput single-cell resolution imaging of nanocarriers, enabling researchers to move from descriptive observations to statistically rigorous, predictive models in drug development.
SCP-Nano research aims to quantify the uptake, trafficking, and fate of therapeutic nanocarriers at the single-cell level. Traditional microscopy-based approaches are limited in the number of cells, conditions, and timepoints they can feasibly analyze, leading to underpowered studies susceptible to cellular heterogeneity. This document outlines integrated strategies to overcome these limitations.
High-Throughput Imaging Platforms & Automation
Automated Liquid Handling and Plating
Robotic systems enable consistent seeding of cells in multi-well plates (96, 384, or 1536-well formats), precise nanocarrier dosing, and parallel staining protocols.
Key Protocol: Automated Cell Seeding & Dosing
- Cell Preparation: Harvest and resuspend cells in growth medium to a standardized density (e.g., 5,000 cells/mL for a 96-well plate).
- Automated Dispensing: Use a liquid handler (e.g., Integra ViaFlo, Beckman Coulter Biomek) to dispense 100 µL/well into a black-walled, glass-bottom microplate.
- Incubation: Allow cells to adhere for 24 hours in a controlled incubator.
- Nanocarrier Dosing: Prepare a serial dilution of fluorescently labeled nanocarriers. The liquid handler dispenses dosing solutions across plate columns, creating a concentration gradient.
- Fixation & Staining: At defined timepoints, an automated plate washer aspirates medium, and the handler adds fixative (4% PFA), permeabilization buffer (0.1% Triton X-100), and counterstains (e.g., DAPI, Phalloidin).
Confocal High-Content Imaging Systems
These systems combine automated microscopy with quantitative image analysis.
Key Platform Parameters:
| Platform | Max Wells/Plate | Z-stack Capability | Live-Cell Imaging | Typical Scan Time (96-well) |
|---|---|---|---|---|
| PerkinElmer Opera Phenix | 1536 | Yes | Yes (environmental control) | ~45 min (2 channels, 9 sites/well) |
| Molecular Devices ImageXpress | 1536 | Yes | Yes (optional) | ~60 min |
| Yokogawa CV8000 | 384 | Yes | Yes | ~30 min |
Computational Pipelines for Image Analysis
Segmentation and Feature Extraction
Cell segmentation is performed using algorithms (e.g., CellPose, StarDist) trained on DAPI and membrane stains. For each cell, dozens of features are extracted.
Table 1: Key Quantitative Features Extracted in SCP-Nano Analysis
| Feature Category | Specific Metrics | Biological Relevance |
|---|---|---|
| Nanocarrier Uptake | Total fluorescence intensity per cell, Integrated puncta count, Mean puncta intensity. | Measures cellular dose. |
| Spatial Distribution | Cytosolic vs. perinuclear intensity ratio, Colocalization coefficients (Manders, Pearson) with organelle markers (Lysotracker, ER tracker). | Indicates subcellular trafficking and potential escape mechanisms. |
| Cell Morphology | Cell area, perimeter, nuclear size, Cytoskeletal texture. | Correlates nanocarrier interaction with cell state. |
| Population Statistics | Coefficient of Variation (CV) in uptake, Skewness of intensity distribution. | Quantifies population heterogeneity. |
Analysis Workflow Diagram
Diagram Title: SCP-Nano High-Content Analysis Computational Pipeline
Experimental Design for Statistical Power
Power Analysis for SCP-Nano Studies
To determine the required number of cells (n) per condition, an a priori power analysis is essential.
Protocol: Power Calculation for Mean Difference in Uptake
- Pilot Study: Run a small-scale experiment (3 replicates) for control and treated groups.
- Calculate Effect Size (d): d = (MeanIntensityTreat - MeanIntensityControl) / Pooled Standard Deviation.
- Set Parameters: Define desired statistical power (1-β, typically 0.8) and significance level (α, typically 0.05).
- Use Software: Input d, α, and power into tools (G*Power, R
pwrpackage) to calculate required n per group. - Scale Experiment: If n=500 cells/group is needed and segmentation yields ~1000 cells/well, then ≥1 well/condition suffices. Include ≥3 biological replicates (different cell passages).
Multiplexed Experimental Design
Barcoding nanocarriers with distinct fluorescent signatures (e.g., different ratios of quantum dots) allows pooling of multiple conditions into one well, controlling for well-to-well variability.
Table 2: Comparison of Standard vs. Barcoded SCP-Nano Design
| Design Aspect | Standard Workflow | Barcoded Multiplex Workflow |
|---|---|---|
| Conditions per Well | 1 | 4-10 (via spectral barcodes) |
| Key Reagent | Single-label nanocarrier | Spectrally barcoded nanocarrier library |
| Throughput Gain | 1x | 4-10x |
| Data Normalization | Cross-well | Intra-well, against internal control cells |
| Main Challenge | Plate uniformity | Unmixing complex spectra, carrier synthesis |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Research Reagent Solutions for SCP-Nano Studies
| Item | Function & Rationale | Example Product/Chemical |
|---|---|---|
| Glass-Bottom Microplates | Provide optimal optical clarity for high-resolution imaging, essential for confocal Z-stacks. | MatriPlate 96-well, #1.5 cover glass bottom. |
| Fluorescent Organelle Reporters | Live-cell or fixed-cell markers to determine nanocarrier colocalization and trafficking pathways. | LysoTracker Deep Red (lysosomes), MitoTracker Green (mitochondria), ER-Tracker Blue-White DPX. |
| Membrane Stains | Enable accurate cytoplasmic segmentation for single-cell analysis. | CellMask Deep Red (live), Wheat Germ Agglutinin (WGA) conjugates (fixed). |
| Fixable Viability Dyes | Distinguish live from dead cells during analysis; fixable for later processing. | Zombie NIR (BioLegend), Fixable Viability Dye eFluor 780. |
| Automated Liquid Handling Tips | Low-retention tips ensure precise, reproducible nanocarrier dosing across a plate. | Beckman Coulter Biomek 3000 P250 Low-Retention Tips. |
| Phenotypic Screening Libraries | Pharmacologic tool compounds to perturb cellular pathways and study their effect on nanocarrier uptake (e.g., endocytosis inhibitors). | Cayman Chemical Endocytosis Inhibitor Library. |
| Spectral Unmixing Reference Dyes | Essential for experiments using barcoded nanocarriers; create a reference spectrum for each barcode. | Sets of individual quantum dots (CdSe/ZnS) at distinct emissions. |
Integrated Signaling Pathway Analysis
Understanding how cell signaling states affect nanocarrier processing is key. High-throughput imaging enables correlation of pathway activation (via biosensors) with uptake.
Diagram Title: Signaling Pathways Influencing Nanocarrier Uptake
Scaling SCP-Nano studies requires a synergistic integration of automated experimental workflows, multiplexed design, and robust computational analysis. By implementing the strategies outlined—leveraging high-content screening, power-aware experimental design, and barcoding—researchers can achieve the throughput and statistical power necessary to decode the complex, heterogeneous interactions between nanocarriers and single cells, ultimately accelerating the rational design of next-generation drug delivery systems.
Benchmarking SCP-Nano: Performance Validation and Comparative Analysis with Alternative Techniques
Within the advancing field of SCP-Nano single-cell resolution nanocarrier imaging research, rigorous validation of quantitative data is paramount. This technical guide details the systematic approach for correlating and validating key quantification metrics obtained from three cornerstone analytical techniques: Flow Cytometry (FC), High-Performance Liquid Chromatography (HPLC), and Mass Spectrometry (MS). The convergence of data from these orthogonal methods establishes a robust framework for verifying nanocarrier uptake, drug payload, and single-cell pharmacokinetic parameters, thereby ensuring the reliability essential for downstream drug development.
SCP-Nano research aims to track, quantify, and understand the behavior of nanocarriers at the individual cell level. Each analytical platform provides complementary but distinct data dimensions. Flow cytometry offers high-throughput, single-cell statistical data on nanocarrier association. HPLC provides precise separation and quantification of molecular species (e.g., released drugs, lipids, polymers) from bulk or lysate samples. Mass spectrometry delivers unparalleled specificity and sensitivity for identifying and quantifying compounds, often in complex matrices. Correlating results across these platforms is the gold standard for validating any single measurement, mitigating instrument-specific artifacts, and building a complete, credible quantification profile.
Core Methodologies and Protocols
Experimental Workflow for Correlative Analysis
Diagram Title: Workflow for Multi-Technique Quantification Validation
Protocol: Integrated Sample Preparation for Tri-Method Analysis
- SCP-Nano Treatment: Incubate target cells (e.g., HEK293, HeLa, primary lymphocytes) with fluorescently labeled nanocarriers (e.g., DiO-labeled liposomes, Cy5-conjugated polymer nanoparticles) at relevant concentrations and time points.
- Sample Splitting: Harvest cells and wash 3x with PBS. Split the single-cell suspension into three aliquots.
- Aliquot A (FC): Resuspend in cold PBS + 1% FBS for immediate flow cytometry.
- Aliquot B (HPLC/MS - Lysate): Pellet cells. Lyse using RIPA buffer or 70/30 methanol/water with 0.1% formic acid. Centrifuge (16,000g, 15 min, 4°C). Collect supernatant for analysis.
- Aliquot C (MS - Direct Injection): For live cell or targeted MS, pellet and resuspend in minimal buffer for direct injection or specialized MS sample prep (e.g., NanoDESI, live-cell probe).
- Parallel Processing: Run all three analytical techniques within a 24-hour window to prevent degradation.
Flow Cytometry Protocol for Nanocarrier Association
Objective: Quantify cell population heterogeneity and mean fluorescence intensity (MFI) as a proxy for nanocarrier uptake/cell association.
- Instrument Calibration: Use calibration beads (e.g., Spherotech Rainbow Beads) daily to ensure PMT stability and inter-day reproducibility.
- Gating Strategy: Record ≥10,000 events per sample. Gate on live, single cells using FSC-A/SSC-A and FSC-H/FSC-W.
- Fluorescence Detection: Detect nanocarrier label (e.g., FITC, DiO: 530/30 nm filter; Cy5: 670/30 nm filter). Use an unstained control to set the negative population threshold.
- Data Output: Export MFI, median fluorescence, and % positive cells. Perform statistical analysis (mean ± SEM) across replicates.
HPLC Protocol for Payload Quantification
Objective: Separate and quantify the encapsulated or released drug from nanocarriers or cell lysates.
- Column: C18 reversed-phase column (e.g., 2.1 x 50 mm, 1.7 µm particle size).
- Mobile Phase: A: 0.1% Formic acid in water; B: 0.1% Formic acid in acetonitrile.
- Gradient: 5% B to 95% B over 8 min, hold 2 min, re-equilibrate.
- Flow Rate: 0.3 mL/min.
- Detection: UV-Vis/DAD (at λmax of target drug) or coupled to MS.
- Quantification: Use a 5-point external standard curve of the pure drug (e.g., 0.1 - 100 µg/mL). Integrate peak areas.
Mass Spectrometry Protocol for Specific Identification & Quantification
Objective: Provide definitive identification and ultra-sensitive quantification of drug, metabolites, or nanocarrier components.
- Ionization: Electrospray Ionization (ESI), positive or negative mode.
- Mass Analyzer: Triple quadrupole (QqQ) for targeted quantification (MRM) or Q-TOF for untargeted identification.
- MS Parameters (Example for Doxorubicin):
- Precursor ion: [M+H]+ m/z 544.1
- Product ions: m/z 397.1 (quantifier), 361.0 (qualifier)
- Collision energy: Optimized for each transition.
- Data Analysis: Use chromatographic software (e.g., Skyline, MassHunter) to integrate MRM peaks. Quantify against a stable isotope-labeled internal standard (e.g., Doxorubicin-d3).
Table 1: Comparison of Analytical Techniques for SCP-Nano Validation
| Parameter | Flow Cytometry | HPLC (UV/DAD) | Mass Spectrometry (QqQ MRM) |
|---|---|---|---|
| Primary Output | Cellular fluorescence, population distributions | Chromatographic peak area/retention time | Ion abundance, m/z ratio |
| Quantifiable Metric | Mean Fluorescence Intensity (MFI), % Positive Cells | Concentration (µg/mL) | Concentration (pg/mL - ng/mL) |
| Sample Throughput | High (1000s cells/sec) | Medium | Low-Medium |
| Sensitivity | Moderate (limited by fluorophore) | Moderate-High (ng) | Very High (pg-fg) |
| Specificity | Low-Moderate (subject to autofluorescence) | Moderate (co-elution possible) | Very High (mass-specific) |
| Key Role in Validation | Confirms cellular association at single-cell level. | Confirms bulk drug quantity, purity. | Confirms drug identity, enables ultra-sensitive & multiplexed quantification. |
| Typical Correlation Target | MFI vs. intracellular drug conc. (from MS) | HPLC peak area vs. MS MRM area | Gold standard for all quantification. |
Table 2: Example Correlation Data from a Model SCP-Nano Study (Liposomal Doxorubicin)
| Cell Line | Flow Cytometry MFI (A.U.) | HPLC (UV) Intracellular Dox (ng/10^6 cells) | MS (MRM) Intracellular Dox (ng/10^6 cells) | Correlation (HPLC vs MS) R² | Correlation (MFI vs MS) R² |
|---|---|---|---|---|---|
| MCF-7 (Wild-type) | 15,240 ± 1,100 | 185.6 ± 12.3 | 179.8 ± 9.7 | 0.992 | 0.874 |
| MCF-7 (Resistant) | 5,670 ± 850 | 62.1 ± 8.9 | 58.4 ± 5.2 | 0.987 | 0.901 |
| HEK293 | 8,450 ± 720 | 105.3 ± 10.1 | 101.5 ± 8.4 | 0.995 | 0.812 |
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Validation Experiments
| Item | Function in Validation | Example Product/Catalog |
|---|---|---|
| Fluorescent Lipophilic Dyes | Label nanocarrier membrane for flow cytometry tracking. | DiO (Invitrogen D275), DiD (Invitrogen D7757) |
| Cell Lysis Buffer (MS-compatible) | Extract drugs and analytes from cells without interfering with MS ionization. | RIPA Buffer (MS-grade), Methanol/Water with Formic Acid |
| Stable Isotope-Labeled Internal Standards | Normalize for MS ionization efficiency and sample preparation losses, enabling absolute quantification. | Doxorubicin-d3, Paclitaxel-d5 |
| HPLC Calibration Standards | Generate external standard curves for HPLC quantification. | Drug master standard (e.g., Sigma-Aldrish D1515 for Doxorubicin) |
| Flow Cytometry Calibration Beads | Standardize instrument fluorescence intensity across experiments and days. | Spherotech Rainbow Calibration Particles (RCP-30-5A) |
| C18 Solid Phase Extraction (SPE) Plates | Clean-up and concentrate cell lysate samples pre-HPLC/MS to remove salts and impurities. | Waters Oasis HLB µElution Plate |
| MS Mobile Phase Additives | Enhance ionization efficiency and chromatographic peak shape in LC-MS. | Optima LC/MS Grade Formic Acid, Ammonium Acetate |
Pathway for Data Integration and Decision Logic
Diagram Title: Decision Logic for Data Validation
The synergistic application of flow cytometry, HPLC, and mass spectrometry creates an irrefutable chain of evidence for quantification in SCP-Nano research. By adhering to the detailed protocols, employing the essential toolkit reagents, and following the structured correlation framework outlined herein, researchers can achieve and demonstrate a level of quantitative rigor that meets the stringent demands of modern therapeutic nanocarrier development. This multi-modal validation strategy transforms single-cell imaging data from qualitative observations into robust, quantitative metrics ready for translational decision-making.
Within the thesis framework of SCP-Nano (Single-Cell Precision Nanocarrier) research, the ability to visualize and quantify nanocarrier delivery, uptake, and payload release at genuine single-cell resolution is paramount. This technical guide analyzes the core performance metrics—spatial resolution and detection sensitivity—of three pivotal imaging modalities: conventional confocal laser scanning microscopy (CLSM), light-sheet fluorescence microscopy (LSFM), and the emerging SCP-Nano imaging platform. The choice of platform directly dictates the granularity of data obtainable in key experiments, from tracking individual nanoparticle binding events to quantifying heterogeneity in intracellular drug release kinetics.
Technical Performance Comparison: Quantitative Metrics
The following tables summarize key performance parameters based on current state-of-the-art systems and published benchmarks.
Table 1: Core Imaging Performance Metrics
| Parameter | Conventional Confocal (CLSM) | Light-Sheet (LSFM/SPIM) | SCP-Nano Platform (Emergent) |
|---|---|---|---|
| Lateral (XY) Resolution | ~240-280 nm (with pinhole) | ~300-400 nm (inherent) | < 100 nm (reported) |
| Axial (Z) Resolution | ~500-700 nm | ~800-1000 nm | ~150-200 nm (reported) |
| Typical Imaging Speed | 0.1 - 2 fps (512x512) | 10 - 200 fps (volume rates) | 1 - 10 fps (super-res) |
| Phototoxicity & Bleaching | High (point scanning) | Very Low (plane illumination) | Moderate (structured illumination) |
| Typical Penetration Depth | ~50-100 µm (cleared samples) | 500 µm - several mm (cleared) | ~50-100 µm (high-NA) |
| Single-Particle Sensitivity | Moderate (SNR limited by pinhole) | Good (high SNR) | Excellent (high SNR + dedicated detectors) |
| Live-Cell Compatibility | Moderate | High | Moderate-High |
Table 2: Performance in SCP-Nano Research Applications
| Application | Confocal Challenge | Light-Sheet Advantage | SCP-Nano Proposed Solution |
|---|---|---|---|
| Nanocarrier Binding & Uptake | Diffraction-limited; cannot resolve sub-diffraction clusters. | Fast 3D tracking but lower resolution. | Quantify binding sites <100 nm apart; count individual carriers. |
| Intracellular Trafficking | Photobleaching limits long 4D tracks. | Excellent for long-term 3D trajectories. | High-res 3D tracks of carriers in organelles. |
| Payload Release (Ratiometric) | Slow acquisition can miss kinetics. | Fast volumetric imaging captures dynamics. | Super-resolved release maps co-localized with organelles. |
| Heterogeneity Mapping | Sectioning induces artifacts in population analysis. | True 3D census of thousands of cells. | High-content, high-resolution single-cell profiling. |
Detailed Experimental Protocols for SCP-Nano Research
Protocol 1: High-Resolution Mapping of Nanocarrier Binding Sites
- Objective: Quantify the density and distribution of targeted SCP-Nano carriers on the plasma membrane of live cells.
- Sample Prep: Incubate cells with SCP-Nano carriers carrying a far-red fluorophore (e.g., Cy5) and a cell mask dye (e.g., CellMask Green). Fix lightly if necessary.
- SCP-Nano Imaging:
- Mount sample in physiological buffer.
- Use TIRF-SIM (Total Internal Reflection Fluorescence - Structured Illumination) mode of the SCP-Nano system.
- Acquire sequential, high-NA TIRF images (488 nm, 640 nm) followed by 3-phase SIM patterns for each channel.
- Reconstruct super-resolution images using proprietary algorithms.
- Apply particle detection and DBSCAN clustering analysis to quantify carrier distribution.
- Comparative Control: Image the same FOV with a high-end confocal (e.g., Airyscan detector) using optimal Nyquist sampling.
Protocol 2: Volumetric Tracking of Intracellular Trafficking & Payload Release
- Objective: Follow single carriers in 3D over time and correlate location with pH-sensitive payload release.
- Sample Prep: Use dual-labeled SCP-Nano carriers: a stable dye (e.g., Alexa Fluor 647) on the particle shell and a pH-sensitive dye (e.g., pHrodo Green) on the encapsulated payload.
- SCP-Nano/LSFM Hybrid Workflow:
- Long-Term 3D Tracking: Use LSFM in multi-view mode to image cells over 12-24 hours, acquiring volumetric stacks every 30 seconds (488 nm, 560 nm excitation). This provides low-photoxicity trajectories.
- High-Resolution Snapshots: At defined timepoints (e.g., 1h, 4h, 12h), pause LSFM, relocate the cell of interest, and switch to the SCP-Nano system's 3D-SIM or confocal mode.
- Acquire high-resolution 3D stacks (xy: ~120 nm, z: ~250 nm) of the cell using both wavelengths.
- Co-register LSFM and SCP-Nano datasets. Use ratiometric analysis (pHrodo/AF647) on the high-res images to pinpoint exact subcellular location (e.g., early endosome vs. lysosome) at the moment of payload release.
Visualization of Methodologies and Pathways
Title: High-Res Nanocarrier Binding Site Mapping Workflow
Title: Intracellular Trafficking and pH-Dependent Release Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for SCP-Nano Imaging Experiments
| Reagent / Material | Function in SCP-Nano Research | Key Consideration |
|---|---|---|
| pH-Sensitive Fluorophores (e.g., pHrodo, SNARF) | Report on endosomal maturation and payload release kinetics via ratiometric or intensity-based signals. | Match pKa to intended compartment (e.g., pHrodo Red for lysosomes). |
| Photostable Dyes (e.g., Alexa Fluor 647, CF680) | Label the nanocarrier shell for long-term, high-resolution tracking without bleaching. | Essential for single-particle tracking (SPT) and super-resolution imaging. |
| Organelle-Specific Dyes (e.g., LysoTracker, MitoTracker) | Provide spatial context for carrier localization within live cells. | Use at low concentration to avoid toxicity and imaging artifacts. |
| Metabolic Labeling Dyes (e.g., HaloTag, SNAP-tag substrates) | For covalent, specific labeling of engineered cellular targets or carrier surfaces. | Enables pulse-chase and orthogonal labeling strategies. |
| Optically Clear Mounting Media / Refractive Index Matchers | Minimize spherical aberration and scattering for deep, high-resolution imaging. | Critical for maintaining resolution in 3D-SIM and confocal modes. |
| Fiducial Markers (e.g., TetraSpeck beads) | For precise channel alignment and drift correction in multi-color, super-res imaging. | Necessary for accurate co-localization analysis. |
| Live-Cell Imaging Buffers (e.g., CO2-independent media, antioxidants) | Maintain cell viability and physiology during long-term, high-intensity imaging sessions. | Reduces oxidative stress and phototoxicity. |
This whitepaper is framed within a broader thesis on SCP-Nano (Single-Cell Precision Nanocarrier) imaging research, a revolutionary modality enabling real-time, single-cell resolution tracking of therapeutic nanocarriers in vivo. For researchers and drug development professionals, selecting the appropriate imaging technology is critical. This guide provides a comparative analysis to inform that decision.
SCP-Nano: Utilizes engineered nanocarriers (e.g., polymeric, lipid-based) tagged with multiplexed fluorophores or rare-earth isotopes. Detection is via highly sensitive, wide-field intravital microscopy or mass cytometry, offering dynamic, molecular-level data at the single-cell level.
Transmission Electron Microscopy (TEM): Employs a beam of electrons transmitted through an ultra-thin specimen to produce high-resolution, nanometer-scale structural images, primarily of fixed samples.
Positron Emission Tomography (PET): A nuclear medicine functional imaging technique that detects gamma rays emitted by a positron-emitting radiotracer to visualize metabolic processes.
Magnetic Resonance Imaging (MRI): Uses strong magnetic fields and radio waves to generate detailed anatomical images based on the magnetic properties of atomic nuclei (e.g., water protons).
Comparative Analysis: Quantitative Metrics
Table 1: Comparative Technical Specifications of Imaging Modalities
| Parameter | SCP-Nano | TEM | PET | MRI |
|---|---|---|---|---|
| Spatial Resolution | 200-500 nm (optical) | 0.05-1 nm | 1-2 mm (clinical) | 10-100 µm (preclinical) |
| Temporal Resolution | Milliseconds to seconds | Minutes to hours (sample prep) | 30 sec to minutes | Minutes to hours |
| Imaging Depth | ~1 mm (intravital) | Thin sections only (<100 nm) | No limit (whole body) | No limit (whole body) |
| Quantitative Output | Single-cell kinetic data, multiplexed target counts | Morphometric data, lattice structures | Picomolar sensitivity to tracer concentration | Relaxometry (T1/T2), contrast agent concentration |
| Throughput | High (live, dynamic tracking) | Very Low (fixed samples) | Moderate (whole-body scans) | Low to Moderate |
Table 2: Application-Specific Suitability
| Research Goal | SCP-Nano | TEM | PET | MRI | Primary Rationale |
|---|---|---|---|---|---|
| Single-Cell Nanocarrier Trafficking | Optimal | Limited | Poor | Poor | Unmatched cellular & subcellular resolution in live tissue. |
| Whole-Body Biodistribution (Macro-scale) | Poor | N/A | Optimal | Optimal | Unlimited depth penetration and field of view. |
| Ultra-structural Cellular Morphology | Limited | Optimal | N/A | Good | Atomic-scale resolution for membrane details, organelles. |
| Real-Time Pharmacokinetics in Live Organism | Optimal | N/A | Good | Limited | High temporal resolution with molecular specificity at cellular scale. |
| Deep-Tissue Hypoxia or Metabolic Profiling | Limited | N/A | Optimal | Good | Sensitivity to picomolar changes in metabolic tracers deep within tissue. |
Detailed Methodologies
Protocol 1: SCP-Nano Intravital Imaging of Liver Tropism
Objective: Quantify cell-specific uptake of targeted lipid nanoparticles (LNPs) in murine liver.
- Nanocarrier Preparation: Formulate LNPs with a PEG-lipid conjugate, ionizable lipid, cholesterol, and a fluorescent dye (e.g., DiR). Conjugate targeting ligand (e.g., galactose) to PEG terminus.
- Animal Preparation: Anesthetize mouse and perform a minimal surgical exposure of the liver lobe. Maintain at 37°C.
- Dosing & Imaging: Administer 2 mg/kg of fluorescent LNP via tail vein. Use a multiphoton microscope with a tunable IR laser (ex: 750 nm, em: 780 nm). Acquire time-lapse images (1 frame/sec for 30 min) of liver sinusoids.
- Data Analysis: Use segmentation software (e.g., Imaris) to distinguish hepatocytes from Kupffer cells based on morphology. Quantify fluorescence intensity per cell over time to generate uptake kinetics curves.
Protocol 2: Correlative TEM Validation of SCP-Nano Findings
Objective: Validate subcellular localization of gold-core nanocarriers imaged via SCP-Nano.
- Sample Fixation: At peak uptake time identified by SCP-Nano, perfuse mouse transcardially with glutaraldehyde (2.5% in cacodylate buffer).
- Embedding & Sectioning: Post-fix in osmium tetroxide, dehydrate, and embed in epoxy resin. Cut 70-nm ultrathin sections.
- Staining & Imaging: Stain sections with uranyl acetate and lead citrate. Image using a 120kV TEM. Identify electron-dense gold nanoparticles within endosomal compartments.
- Correlation: Overlay TEM micrographs with SCP-Nano fluorescence maps using fiduciary markers to confirm organelle-level localization.
Visualizing the SCP-Nano Workflow and Pathway
Title: SCP-Nano Experimental Workflow
Title: Key Intracellular Pathway for SCP-Nano Tracking
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for SCP-Nano Research
| Reagent/Material | Function & Role in Research |
|---|---|
| Ionizable Cationic Lipid (e.g., DLin-MC3-DMA) | Core component of LNPs; enables encapsulation of nucleic acid payloads and endosomal escape. |
| PEG-Lipid (e.g., DMG-PEG2000) | Provides "stealth" properties, modulates pharmacokinetics and prevents aggregation. |
| Near-Infrared Fluorophore (e.g., DiR, Cy7) | High-wavelength dye for deep-tissue, low-background intravital imaging. |
| Heterobifunctional PEG Linker (e.g., MAL-PEG-NHS) | Conjugates targeting ligands (antibodies, peptides) to the nanocarrier surface. |
| Tissue Clearing Agents (e.g., CUBIC, ScaleS) | Renders tissues optically transparent for deep, high-resolution ex vivo validation imaging. |
| Lanthanide Isotope Tags (e.g., 169Yb, 175Lu) | Stable metal isotopes for multiplexed, quantitative detection via mass cytometry (CyTOF). |
The choice between SCP-Nano, TEM, PET, and MRI is dictated by the specific research question. SCP-Nano is the unequivocal choice when the research thesis demands understanding nanocarrier behavior at the single-cell level in a live, dynamic context. It sacrifices imaging depth for unparalleled cellular resolution and kinetic data. TEM provides ultrastructural validation but is static and ex vivo. PET and MRI excel at whole-body, deep-tissue biodistribution but lack the resolution to discern cellular heterogeneity. A synergistic, multi-modal approach, using PET/MRI for macro-distribution and SCP-Nano for micro-mechanisms, represents the future of quantitative nanomedicine research.
This case study is embedded within the broader thesis of Single-Cell Profiling via Nanocarrier (SCP-Nano) imaging research. The core thesis posits that the next generation of effective oncology therapeutics requires nanocarriers engineered not just for tumor accumulation, but for deep, uniform, and single-cell-resolution penetration within the complex tumor microenvironment (TME). Validating such a penetration profile necessitates a multi-modal, cross-platform approach, as no single imaging technique provides a complete spatial and quantitative picture. This guide details the integrated experimental workflow and analytical framework for this essential validation.
Research Reagent Solutions Toolkit
The following table details key reagents and materials essential for the cross-platform validation workflow.
| Item Name | Function in Validation |
|---|---|
| SCP-Nano Construct | Core nanocarrier (e.g., polymeric micelle, liposome) co-loaded with a cytotoxic drug (e.g., Doxorubicin), a near-infrared (NIR) fluorophore (e.g., Cy5.5), and a MRI contrast agent (e.g., Gd-DOTA). Enables multi-modal tracking. |
| 3D Multicellular Tumor Spheroid (MCTS) | In vitro avascular tumor model with physiological barriers (dense extracellular matrix, nutrient/oxygen gradients). Used for high-resolution, quantitative penetration analysis. |
| Orthotopic Tumor-Bearing Mouse Model | In vivo model where cancer cells are implanted in the organ/tissue of origin. Provides the most physiologically relevant TME for penetration studies, including vascularization and stromal components. |
| Matrigel Basement Membrane Matrix | Used to embed spheroids or create in vivo implantation scaffolds to model the extracellular matrix barrier for diffusion studies. |
| Click-iT Plus EdU Cell Proliferation Kit | Enables detection of proliferating cells within tumor sections via bioorthogonal chemistry. Correlates nanocarrier penetration zones with proliferative regions. |
| Anti-CD31 Antibody | Labels endothelial cells for immunohistochemistry. Used to map tumor vasculature and assess carrier proximity to blood vessels (perivascular vs. distal delivery). |
| CellTrace Far Red Cell Proliferation Kit | Fluorescent dye for in vitro pre-labeling of tumor cells in spheroids to delineate cell boundaries for co-localization analysis with nanocarrier signal. |
Experimental Protocols for Cross-Platform Validation
Protocol 3.1: High-Resolution Spheroid Penetration Analysis
Objective: Quantify nanocarrier penetration depth and distribution in a 3D in vitro tumor model. Methodology:
- Generate MCTS (~500 µm diameter) from target cancer cell lines using the hanging-drop or ultra-low attachment plate method.
- Incubate mature spheroids with fluorescently labeled SCP-Nano constructs (e.g., 10 µM equivalent drug concentration) for 1, 4, 12, and 24 hours.
- At each time point, wash spheroids, fix with 4% PFA, and mount for confocal microscopy.
- Acquire z-stack images through the entire spheroid (e.g., 10 µm step size).
- Analysis: Use ImageJ/FIJI to plot fluorescence intensity versus radial distance from the spheroid rim to the core. Calculate the Penetration Index (PI) = (Area under the intensity-depth curve for test sample) / (Area for a superficial, non-penetrating control).
Protocol 3.2: Multi-ModalIn VivoImaging
Objective: Correlate macroscopic tumor accumulation with micro-scale distribution. Methodology:
- Administer the multi-modal SCP-Nano (loaded with NIR dye and MRI agent) intravenously to orthotopic tumor-bearing mice.
- At predetermined times (e.g., 6, 24, 48 h), perform in vivo imaging:
- Fluorescence Molecular Tomography (FMT) or IVIS: Quantify whole-tumor nanocarrier accumulation (as % injected dose/g).
- Magnetic Resonance Imaging (MRI): Acquire T1-weighted maps to visualize heterogeneity of carrier distribution within the tumor.
- Euthanize animals, excise tumors, and snap-freeze in OCT compound for cryosectioning.
Protocol 3.3: Ex Vivo Digital Spatial Profiling of Penetration
Objective: Achieve single-cell resolution data on nanocarrier localization relative to TME features. Methodology:
- Section frozen tumors (10 µm thickness) and stain using multiplex immunofluorescence (e.g., Opal system).
- Panel: DAPI (nuclei), Anti-CD31 (vasculature), Anti-α-SMA (fibroblasts), Anti-pan-cytokeratin (tumor cells), and direct NIR fluorescence from the SCP-Nano.
- Image slides using a multispectral confocal or digital slide scanner.
- Analysis: Utilize image analysis software (e.g., HALO, QuPath) to segment the tumor into concentric zones from blood vessels or the tumor periphery. Quantify the NIR signal intensity per cell within each zone. Perform spatial statistics (e.g., Ripley's K-function) to determine if carrier localization is clustered, random, or dispersed.
Table 1: Multi-Platform Penetration Metrics for SCP-Nano vs. Standard Nanoformulation (SNF)
| Platform / Metric | SCP-Nano (24h) | Standard Nanoformulation (24h) | Measurement Technique |
|---|---|---|---|
| 3D Spheroid Penetration Index (PI) | 0.85 ± 0.07 | 0.45 ± 0.12 | Confocal Microscopy Z-stack Analysis |
| Max Penetration Depth in Spheroid (µm) | 220 ± 15 | 120 ± 30 | Confocal Microscopy Radial Plot |
| In Vivo Tumor Accumulation (%ID/g) | 8.2 ± 1.5 | 7.8 ± 1.8 | FMT/IVIS Imaging |
| MRI Heterogeneity Index (T1 Signal Std. Dev.) | 15.2 ± 3.1 | 28.7 ± 5.6 | T1-weighted MRI Analysis |
| % of Tumor Cells >50µm from Vasculature with Signal | 65% ± 8% | 22% ± 10% | Multiplex Immunofluorescence Spatial Analysis |
| Spatial Co-localization (Manders' Coefficient) with Cancer Cells | 0.78 ± 0.05 | 0.35 ± 0.09 | Digital Image Analysis |
Visualization of Workflow and Relationships
Cross-Platform Validation Workflow Diagram
Barriers to Tumor Penetration & SCP-Nano Goal
Establishing Standardization and Reproducibility Guidelines for the Field
The rapid advancement of SCP-Nano (Single-Cell Profiling via Nanocarrier) imaging research promises unprecedented resolution of drug delivery dynamics and cellular responses. However, the field's growth is hampered by significant inter-laboratory variability in nanocarrier characterization, imaging protocols, and data analysis. This whitepaper, framed within a broader thesis to establish SCP-Nano as a robust, predictive tool in drug development, provides concrete technical guidelines to ensure standardization and reproducibility. These principles are critical for translating single-cell nanocarrier data into reliable insights for therapeutic development.
Foundational Concepts Requiring Standardization
Standardization must begin with the precise definition and characterization of inputs. Key parameters are summarized below.
Table 1: Mandatory Characterization Parameters for SCP-Nano Research
| Parameter | Target Specification (Example) | Recommended Analytical Method | Purpose in Reproducibility |
|---|---|---|---|
| Nanocarrier Size (Hydrodynamic Diameter) | 105.5 ± 3.2 nm | Dynamic Light Scattering (DLS), nanoparticle tracking analysis (NTA) | Defines biodistribution & cellular uptake pathways. |
| Surface Charge (Zeta Potential) | -25.8 ± 2.1 mV | Electrophoretic Light Scattering | Predicts colloidal stability and protein corona formation. |
| Drug Loading Efficiency (DLE) | 88.7% ± 4.1% | HPLC-UV/Vis following carrier disruption | Ensures consistent bioactive payload delivery. |
| Surface Ligand Density | 45 ± 5 ligands per particle | Fluorescence assay, mass spectrometry | Standardizes targeted interaction with cell receptors. |
| Single-Cell Imaging Resolution | ≤ 250 nm lateral | Confocal/Super-resolution microscopy calibration | Enables comparable subcellular localization data. |
Standardized Experimental Protocols
Protocol: Quantitative Single-Cell Uptake via Flow Cytometry
This protocol measures population-level nanocarrier association/uptake.
- Cell Seeding: Seed adherent cells (e.g., HeLa, HUVEC) at 2.5 x 10^4 cells/well in a 24-well plate. Culture for 24h to achieve ~80% confluence.
- Dosing: Apply fluorescently labelled nanocarriers at a standardized particle-per-cell ratio (e.g., 10,000:1) in serum-free medium. Include a no-nanocarrier control.
- Incubation: Incubate at 37°C, 5% CO2 for a precise duration (e.g., 2h).
- Quenching & Harvest: Remove media. Rinse cells 3x with cold PBS containing 0.5 mg/mL heparin (to quench surface-bound signal). Detach cells using a mild, non-enzymatic buffer (e.g., 2 mM EDTA in PBS).
- Analysis: Resuspend cells in cold PBS with propidium iodide (PI) for viability gating. Analyze using a flow cytometer. Report median fluorescence intensity (MFI) of the live cell population (PI-negative) for ≥10,000 events. Normalize to control MFI.
Protocol: High-Resolution Co-localization Imaging via Confocal Microscopy
This protocol standardizes imaging for subcellular fate.
- Sample Preparation: Seed cells on #1.5 glass-bottom dishes. Treat with nanocarriers as in 3.1.
- Fixation & Staining: Fix with 4% paraformaldehyde (15 min), permeabilize with 0.1% Triton X-100 (10 min), and block with 3% BSA (1h). Stain with organelle-specific markers (e.g., anti-LAMP1 for lysosomes, MitoTracker for mitochondria) and counterstain nuclei with DAPI.
- Image Acquisition: Using a calibrated confocal microscope, acquire Z-stacks (0.5 µm step) with identical laser power, gain, and pinhole settings across all experiments. Use sequential scanning to avoid bleed-through.
- Analysis: Use standardized software (e.g., ImageJ/Fiji with Coloc2 plugin) to calculate Manders' overlap coefficients (M1, M2) for nanocarrier signal with each organelle marker. Report thresholds determined using control images.
Visualizing Core Pathways and Workflows
Standardized SCP-Nano Experimental Workflow
Key Nanocarrier Intracellular Trafficking Pathways
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for SCP-Nano Experiments
| Item | Function & Purpose in Standardization |
|---|---|
| NIST-Traceable Nanoparticle Size Standards | Calibrates DLS/NTA instruments to ensure accurate particle size distribution reporting across labs. |
| Fluorescent Lipid/Polymers (e.g., DiD, Cy5-PLGA) | Provides consistent, stable labeling of nanocarriers for tracking; must report dye:carrier ratio. |
| Stable Cell Line with Fluorescent Organelle Markers | (e.g., Cell line expressing GFP-LAMP1). Enables consistent co-localization studies without variable antibody staining. |
| Standardized Fetal Bovine Serum (FBS) Batch | Use a single, large lot for all experiments to minimize variability in protein corona formation. |
| Matched Isotype Control & Validation Antibodies | Critical for accurate flow cytometry and immunofluorescence quantification of cell surface targets. |
| Image Resolution & Calibration Slides | (e.g., sub-diffraction limit fluorescent beads). Verifies imaging system performance pre-experiment. |
| Data & Metadata Schema Template (e.g., adapted from MIACA) | Ensures all experimental parameters (Table 1) are systematically recorded alongside raw data. |
Data Reporting & Metadata Standards
A minimum dataset must accompany all publications. This includes all parameters from Table 1, exact protocols (section 3), analysis software with version and key settings, and raw data access in a public repository (e.g., BioStudies, Zenodo). Adopting the FAIR (Findable, Accessible, Interoperable, Reusable) data principles is mandatory for the field's maturation.
The implementation of these guidelines for material characterization, protocol execution, data acquisition, and reporting is non-negotiable for establishing SCP-Nano single-cell imaging as a reproducible and credible pillar of next-generation drug development. Collective adherence will transform the field from producing fascinating observations to generating robust, predictive knowledge.
Conclusion
SCP-Nano imaging represents a transformative leap in nanomedicine, moving the field from population-averaged data to the critical single-cell and sub-cellular scale. By mastering its foundational principles, implementing robust methodologies, overcoming practical challenges, and rigorously validating findings against gold standards, researchers can unlock unprecedented insights into nanocarrier behavior. This capability is poised to de-risk drug development, rationalize nanocarrier design, and ultimately accelerate the translation of more effective, targeted therapies into the clinic. The future lies in integrating SCP-Nano with multi-omics approaches and AI-driven image analysis to build predictive, high-resolution models of drug delivery in complex biological systems.