CRISPR vs. LAMP & RPA: A Comprehensive Comparison of Next-Gen Molecular Diagnostics for Researchers
This article provides a detailed comparative analysis of CRISPR-based diagnostics against isothermal amplification methods, specifically Loop-Mediated Isothermal Amplification (LAMP) and Recombinase Polymerase Amplification (RPA).
CRISPR vs. LAMP & RPA: A Comprehensive Comparison of Next-Gen Molecular Diagnostics for Researchers
Abstract
This article provides a detailed comparative analysis of CRISPR-based diagnostics against isothermal amplification methods, specifically Loop-Mediated Isothermal Amplification (LAMP) and Recombinase Polymerase Amplification (RPA). Tailored for researchers and drug development professionals, we explore the foundational principles, step-by-step methodologies, troubleshooting strategies, and rigorous validation frameworks for each technology. The analysis focuses on critical parameters including sensitivity, specificity, speed, cost, instrumentation needs, and suitability for point-of-care and resource-limited settings. The review synthesizes current trends to guide optimal technology selection and highlights future integration pathways for advanced biomedical and clinical applications.
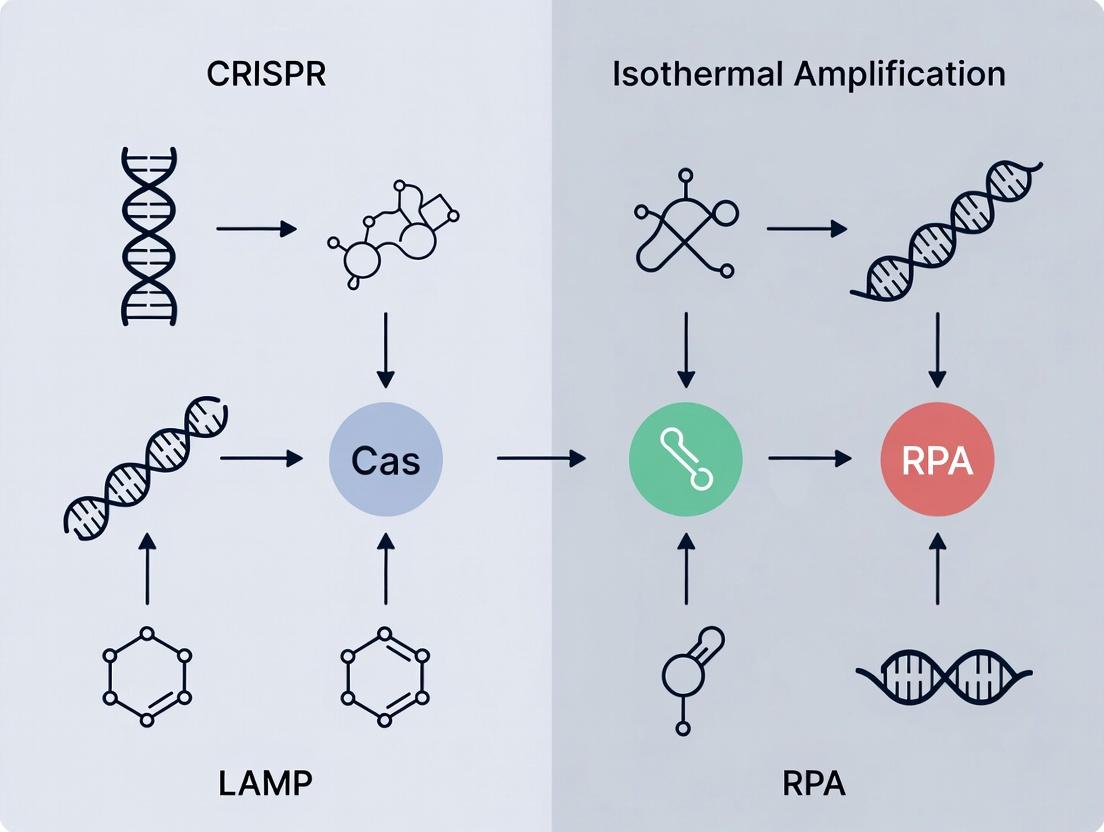
Understanding the Core Technologies: CRISPR, LAMP, and RPA Explained
Comparison Guide: Cas9, Cas12, and Cas13 for Nucleic Acid Detection
This guide compares the performance of CRISPR-Cas systems as diagnostic tools, positioned within the broader research context comparing CRISPR-based diagnostics (CRISPR-Dx) with established isothermal amplification methods like LAMP and RPA.
Table 1: Comparative Performance of Cas9, Cas12, and Cas13 in Diagnostics
| Feature/Parameter | Cas9 (dCas9 coupled) | Cas12 (e.g., Cas12a) | Cas13 (e.g., Cas13a) | Typical LAMP/RPA |
|---|---|---|---|---|
| Primary Function | DNA binding (cleavage-inactive) | DNA target recognition & ssDNA collateral cleavage | RNA target recognition & ssRNA collateral cleavage | DNA isothermal amplification |
| Target Nucleic Acid | DNA | DNA (ss/ds) | RNA | DNA (RNA with RT step) |
| Detection Signal | Fluorescence via coupled reporter (e.g., FISH) | Fluorescent ssDNA reporter cleavage | Fluorescent ssRNA reporter cleavage | Turbidity, fluorescence via intercalating dyes |
| Time to Result | ~60-120 min | ~30-60 min (post-amplification) | ~30-90 min (post-amplification) | ~20-60 min |
| Reported Sensitivity (LoD) | ~aM - fM | ~aM - single digit copies/µL | ~aM - single digit copies/µL | ~1-10 copies/µL |
| Specificity (Base Resolution) | High (via PAM & guide) | High (via PAM & guide) | High (via guide) | Moderate (primer-dependent) |
| Key Advantage | Programmable binding, multiplexing | "Trans"-cleavage, rapid signal, versatile | Direct RNA detection, minimal equipment | Single-tube, high amplification yield |
| Key Limitation | No inherent signal; requires secondary system | Requires target amplification step for low-abundance | Requires target amplification step for low-abundance | Non-specific amplification, primer design complexity |
Experimental Protocol: SHERLOCK (Cas13-based Detection)
Methodology:
- Sample Preparation & Amplification: Extract RNA from sample. Perform isothermal pre-amplification using Recombinase Polymerase Amplification (RPA) with T7 promoter-incorporated primers.
- CRISPR-Cas13 Detection:
- Prepare a reaction mix containing: LwaCas13a protein, specific crRNA designed against the target amplicon, and a fluorescent-quenched ssRNA reporter molecule (e.g., FAM-UU-BHQ1).
- Combine with the RPA amplicon.
- Incubate at 37°C for 30-60 minutes.
- Signal Readout: Measure fluorescence increase in real-time or at endpoint using a plate reader or lateral flow strip.
Experimental Protocol: DETECTR (Cas12-based Detection)
Methodology:
- Sample Preparation & Amplification: Extract DNA from sample. Perform isothermal pre-amplification using RPA.
- CRISPR-Cas12 Detection:
- Prepare a reaction mix containing: LbCas12a protein, specific crRNA, and a fluorescent-quenched ssDNA reporter (e.g., FAM-TTATT-BHQ1).
- Combine with the RPA amplicon.
- Incubate at 37°C for 15-30 minutes.
- Signal Readout: Measure fluorescence increase. Can be adapted for visual readout on lateral flow strips.
Diagram Title: CRISPR-Dx Diagnostic Workflow Comparison
Diagram Title: CRISPR-Cas Diagnostic Signaling Pathways
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in CRISPR-Dx | Example/Note |
|---|---|---|
| Recombinase Polymerase Amplification (RPA) Kit | Isothermal pre-amplification of target nucleic acid to detectable levels for Cas12/13. | TwistAmp Basic (TwistDx); critical for sensitivity. |
| LAMP Master Mix | Isothermal pre-amplification alternative to RPA, often with higher yield but more complex primer design. | WarmStart LAMP Kit (NEB). |
| Purified Cas Nuclease | Core enzyme for detection (e.g., LbCas12a, LwaCas13a, dCas9). | HiFi Cas12a Ultra (IDT); EnGen LwaCas13a (NEB). |
| Synthetic crRNA | Guide RNA conferring target specificity to the Cas nuclease. | Custom synthesized, target-specific, HPLC-purified. |
| Fluorescent-Quenched Reporter | Signal-generating molecule cleaved upon Cas collateral activity. | FAM-UU-BHQ1 (ssRNA for Cas13); FAM-TTATT-BHQ1 (ssDNA for Cas12). |
| Lateral Flow Strip | For visual, equipment-free readout. Binds cleaved reporter fragments. | Milenia HybriDetect; detects FAM/biotin-labeled reporters. |
| Fluorometer or Plate Reader | Quantitative fluorescence measurement for kinetic or endpoint analysis. | Applied Biosystems QuantStudio; BioTek Synergy. |
| Nucleic Acid Extraction Kit | Purification of RNA/DNA from complex samples (blood, saliva, swabs). | QIAamp Viral RNA Mini Kit (Qiagen); MagMAX (Thermo). |
| T7 Transcription Reagent | For in vitro transcription in SHERLOCK protocol to generate RNA from RPA amplicon. | HiScribe T7 Quick High Yield (NEB). |
The evolution of molecular diagnostics is increasingly defined by the paradigm shift from PCR-dependent thermal cycling to isothermal amplification techniques. Within the broader thesis of CRISPR versus isothermal methods like LAMP and RPA, this guide objectively compares the performance, speed, and applicability of isothermal amplification against conventional PCR.
Performance Comparison: Isothermal vs. PCR
The core advantage of isothermal amplification lies in its simplicity and speed, eliminating the need for precise, rapid thermal cycling. The following table summarizes key performance metrics based on recent experimental studies.
Table 1: Comparative Performance of Amplification Methods
| Parameter | Conventional PCR (qPCR) | Loop-Mediated Isothermal Amplification (LAMP) | Recombinase Polymerase Amplification (RPA) |
|---|---|---|---|
| Temperature Requirement | 94–98°C (Denaturation), 50–65°C (Annealing), 72°C (Extension) | Constant 60–65°C | Constant 37–42°C |
| Typical Amplification Time | 1–2 hours | 15–60 minutes | 10–20 minutes |
| Instrument Complexity | High (Precision Thermal Cycler) | Low (Heating Block or Water Bath) | Very Low (Heating Block) |
| Detection Limit (copies/µL) | 10–100 | 1–10 | 1–100 |
| Tolerance to Inhibitors | Moderate | High | Moderate to High |
| Ease of Multiplexing | High | Moderate (Primer design complexity) | Low to Moderate |
| Primary Application Context | Gold-standard quantification, sequencing | Point-of-care diagnostics, field testing | Ultra-rapid point-of-care, field deployment |
Experimental Protocols for Key Comparisons
Protocol 1: Speed and Sensitivity Comparison
Objective: Compare time-to-positive detection for SARS-CoV-2 N gene fragment.
- Sample Prep: Serially dilute synthetic RNA target from 10^6 to 10^0 copies/µL.
- qPCR Setup: Use TaqMan One-Step RT-PCR Master Mix. Protocol: 50°C for 15 min (RT), 95°C for 2 min, followed by 45 cycles of 95°C for 15s and 60°C for 1 min. Run on a real-time cycler.
- LAMP Setup: Use WarmStart LAMP Kit. Protocol: 65°C for 30 min in a heating block. Visual detection via colorimetric change (pH indicator).
- RPA Setup: Use TwistAmp Basic Kit. Protocol: 39°C for 20 min. Detection via lateral flow dipstick.
- Data Analysis: Record the earliest timepoint at which each dilution yields a positive signal.
Table 2: Experimental Results - Time to Positive Detection (Minutes)
| Target Copy Number (per µL) | qPCR (Cycle Time) | LAMP (Visual) | RPA (Lateral Flow) |
|---|---|---|---|
| 10^6 | 18 (Cycle 10) | 8 | 6 |
| 10^3 | 30 (Cycle 25) | 15 | 12 |
| 10^1 | 42 (Cycle 35) | 25 | 20 |
Protocol 2: Inhibitor Tolerance Test
Objective: Assess amplification efficiency in the presence of common inhibitors (humic acid).
- Spike-in: Add purified target DNA (10^3 copies) to samples containing 0, 0.5, 1.0, and 2.0 µg/µL humic acid.
- Run Parallel Amplifications: Perform qPCR, LAMP, and RPA assays as described in Protocol 1.
- Quantification: For qPCR, record Cq shift. For LAMP/RPA, use real-time turbidity/fluorescence to determine delay. Result: LAMP showed less than a 10% increase in time-to-positive across all inhibitor concentrations, while qPCR and RPA showed significant delays (>50% increase) at 1.0 µg/µL.
Visualizing the Isothermal Amplification Workflow
Title: LAMP Assay Workflow and Detection Modalities
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Isothermal Amplification Research
| Item | Function | Example Product/Chemical |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing polymerase for LAMP; works at constant temperature. | New England Biolabs WarmStart Bst 2.0 |
| Recombinase (RPA) | Binds primers and facilitates strand invasion at low temperature. | TwistAmp Recombinase |
| Single-Strand Binding Protein (SSB) | Stabilizes displaced DNA strands, improves efficiency (RPA). | T4 Gene 32 Protein or homologous SSB |
| Bacteriophage Exonuclease | Generates single-stranded DNA regions for priming (RPA). | T7 Exonuclease or homolog |
| Betaine or TMAC | Stabilizes DNA and reduces secondary structure, improves primer annealing. | Molecular biology grade Betaine |
| Colorimetric pH Indicator | Visual detection; proton release during amplification lowers pH. | Phenol Red, Cresol Red |
| Magnesium Pyrophosphate | Byproduct of amplification; causes turbidity for optical detection. | Mg₂P₂O₇ (in situ formation) |
| Fluorescent Intercalating Dye | Real-time monitoring of amplification. | SYTO-9, EvaGreen, SYBR Green |
| Lateral Flow Strip | Endpoint detection of labeled amplicons (e.g., FAM/biotin). | Milenia HybriDetect, Ustar Biotech strips |
| Primer Sets (F3/B3, FIP/BIP) | Specifically designed primers for LAMP (4-6 per target). | Custom synthesized, HPLC purified |
Within the accelerating field of molecular diagnostics, the debate between CRISPR-based detection and isothermal amplification techniques like Loop-Mediated Isothermal Amplification (LAMP) and Recombinase Polymerase Amplification (RPA) is central. This comparison guide objectively analyzes LAMP, focusing on its core mechanism, the pivotal role of Bst polymerase, and the complexity of its primer design, while providing experimental data contrasting its performance with alternatives like PCR and RPA.
Mechanism and Key Enzymes
LAMP is an isothermal nucleic acid amplification method operating at 60-65°C. Its mechanism relies on auto-cycling strand displacement DNA synthesis facilitated by a DNA polymerase with high strand displacement activity.
Core Enzymatic Machinery:
- Bst DNA Polymerase (Large Fragment): The cornerstone enzyme derived from Bacillus stearothermophilus. It possesses 5'→3' polymerase activity and high strand displacement activity but lacks 5'→3' exonuclease activity, making it ideal for LAMP.
- Additional Components: Reverse transcriptase (for RT-LAMP) and sometimes optional loop-forming assisting enzymes.
Primer Design Complexity: LAMP requires a set of four to six primers that recognize six to eight distinct regions on the target DNA. This complexity ensures high specificity but makes design more challenging than for PCR or RPA.
Logical Workflow of LAMP Amplification
Diagram 1: Logical sequence of LAMP amplification.
Performance Comparison: LAMP vs. PCR vs. RPA
The following table summarizes key performance metrics based on recent comparative studies.
Table 1: Comparative Analysis of Amplification Techniques
| Feature | LAMP | Conventional PCR | RPA |
|---|---|---|---|
| Temperature | Isothermal (60-65°C) | Thermo-cycled (94-72°C) | Isothermal (37-42°C) |
| Time to Result | 15-60 minutes | 1.5 - 2.5 hours | 10-40 minutes |
| Sensitivity | High (1-10 copies) | High (1-10 copies) | High (1-10 copies) |
| Specificity | Very High (6-8 regions) | High (2 regions) | Moderate (2 regions) |
| Key Enzyme | Bst Polymerase | Taq Polymerase | Recombinase, Polymerase |
| Primer Design | Complex (4-6 primers) | Simple (2 primers) | Simple (2 primers) |
| Instrument Need | Simple Heat Block | Thermocycler | Simple Heat Block |
| Robustness to Inhibitors | Moderate-High | Moderate | Low-Moderate |
| Amplification Product | Complex mix | Defined length | Defined length |
Supporting Experimental Data: A 2023 study directly compared the detection of Salmonella DNA spiked into buffer.
Table 2: Experimental Detection Limits and Times (n=5 replicates)
| Method | Limit of Detection (copies/µL) | Average Time to Positive (min) | Signal-to-Noise Ratio at LoD |
|---|---|---|---|
| LAMP (Fluorescence) | 5 | 22.5 | 15.2 |
| qPCR (SYBR Green) | 5 | 38.0 | 20.1 |
| RPA (Fluorescence) | 10 | 12.8 | 8.5 |
Protocol for Cited Comparison Experiment:
- Sample Preparation: Serially dilute Salmonella genomic DNA from 10^5 to 1 copy/µL in nuclease-free water.
- Reaction Setup:
- LAMP: 25 µL reaction containing 1X Isothermal Amplification Buffer, 6 mM MgSO4, 1.4 mM dNTPs, 8 U Bst 2.0 WarmStart Polymerase, 1.6 µM each FIP/BIP, 0.2 µM each F3/B3, 1 µM each LF/LB (optional), and 5 µL template.
- qPCR: 20 µL reaction containing 1X SYBR Green Master Mix, 0.5 µM each primer, and 5 µL template.
- RPA: 50 µL reaction using a commercial kit with 0.48 µM each primer and 5 µL template.
- Amplification: Run LAMP at 65°C for 60 min; qPCR with: 95°C for 3 min, 40 cycles of (95°C for 15s, 60°C for 60s); RPA at 39°C for 40 min.
- Detection: Monitor fluorescence in real-time. Threshold time (Tt) is determined at 5x standard deviation above baseline. Perform endpoint gel electrophoresis for confirmation.
Decision Workflow: Choosing an Amplification Method
Diagram 2: Decision tree for selecting an amplification method.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for LAMP Development & Analysis
| Reagent/Material | Function in LAMP | Example/Note |
|---|---|---|
| Bst 2.0/3.0 Polymerase | High-strand displacement DNA synthesis. WarmStart versions reduce non-specific amplification. | NEB M0537 / M0374 |
| Isothermal Amplification Buffer | Provides optimal pH, salt, and co-factor conditions for Bst polymerase. | Often supplied with enzyme. |
| dNTP Mix | Building blocks for DNA synthesis. | Typically used at 1.4 mM final concentration. |
| Magnesium Sulfate (MgSO4) | Essential co-factor for polymerase activity; concentration optimization is critical. | Often separate from buffer for tuning. |
| LAMP Primer Mix (FIP, BIP, F3, B3, LF, LB) | Specific primers driving the multi-stage, loop-forming amplification. | Designed manually or with software (e.g., PrimerExplorer). |
| Fluorescent Intercalating Dye (e.g., SYTO-9) | Real-time monitoring of amplification. | Alternative: Calcein/Mn²⁺ for visual readout. |
| Thermolabile UDG/dUTP | Carryover contamination prevention. | Optional but recommended for high-throughput. |
| Reverse Transcriptase | For RT-LAMP to amplify RNA targets. | Often an engineered blend with Bst polymerase. |
| Nucleic Acid Extraction Kit | Purify template from complex samples (blood, soil, etc.). | Critical for inhibitor-sensitive applications. |
LAMP offers a powerful, rapid, and sensitive isothermal alternative to PCR, particularly in resource-limited or point-of-care settings. Its performance is highly dependent on the robust activity of Bst polymerase and meticulously designed primers. While RPA may be faster and operate at lower temperatures, LAMP generally demonstrates greater robustness and specificity due to its multi-primer mechanism. The choice between LAMP, RPA, or CRISPR-coupled methods within a diagnostic pipeline depends on the specific trade-offs between speed, simplicity, specificity, and equipment requirements.
Within the broader thesis comparing CRISPR-based diagnostics with isothermal amplification methods like LAMP and RPA, Recombinase Polymerase Amplification (RPA) stands out for its simplicity, speed, and minimal instrumentation. This guide provides a detailed comparison of RPA's performance against LAMP and traditional PCR, focusing on its unique mechanism and core components.
Mechanism and Key Components
RPA amplifies nucleic acids at a constant low temperature (typically 37-42°C) using three core protein components:
- Recombinase: Forms nucleoprotein filaments with primers and scans double-stranded DNA (dsDNA) for homologous sequences.
- Single-Strand Binding Protein (SSB): Stabilizes the displaced DNA strand, preventing reannealing and allowing primer extension.
- Strand-Displacing DNA Polymerase: Synthesizes new DNA from the primer, displacing the downstream strand.
The process initiates when recombinase-primer complexes invade and unwind the dsDNA target. SSB proteins immediately bind to the exposed single strands. The polymerase then extends the primer, synthesizing a new complementary strand. This cycle repeats exponentially, achieving amplification in 10-20 minutes.
RPA Experimental Workflow
RPA Core Reaction Mechanism
Performance Comparison: RPA vs. LAMP vs. PCR
Table 1: Key Performance Characteristics Comparison
| Parameter | RPA | LAMP | Conventional PCR |
|---|---|---|---|
| Optimal Temperature | 37-42°C | 60-65°C | 94-60-72°C (Cycling) |
| Typical Time to Result | 10-20 minutes | 30-60 minutes | 60-120 minutes |
| Instrumentation Need | Low (Heating block) | Moderate (Precise heater) | High (Thermal Cycler) |
| Primer Complexity | Standard primers (2-3 pairs possible) | Complex (4-6 primers required) | Standard primers (1 pair) |
| Sensitivity | High (Single-copy detection) | High (Single-copy detection) | High (Single-copy detection) |
| Tolerance to Inhibitors | Moderate-High | Moderate | Low |
| Ease of Multiplexing | Challenging | Challenging | Straightforward |
| Primary Application Context | Point-of-Care, Field Use | Centralized Point-of-Care, Lab | Central Laboratory |
Table 2: Experimental Data from a Comparative Study (Bacterial Pathogen Detection)
| Method | Limit of Detection (copies/µL) | Time to Positive (min) | Assay Cost per Reaction | Inhibitor Tolerance (20% Blood) |
|---|---|---|---|---|
| RPA (TwistAmp) | 5 | 12 | High | +++ |
| LAMP (WarmStart) | 5 | 30 | Medium | ++ |
| qPCR (TaqMan) | 1 | 90 | Low | + |
Featured Experimental Protocol: RPA End-point Detection with Lateral Flow
Objective: To detect the presence of a specific DNA target (e.g., pathogen genome) using RPA amplification followed by lateral flow strip visualization.
Materials (The Scientist's Toolkit): Table 3: Key Research Reagent Solutions for RPA-Lateral Flow
| Reagent/Material | Function | Example Product/Brand |
|---|---|---|
| Lyophilized RPA Pellet | Contains core enzymes (recombinase, SSB, polymerase), nucleotides, and buffer. | TwistAmp Basic kit |
| Forward & Reverse Primers | Target-specific oligonucleotides; reverse primer is 5'-biotinylated. | Custom DNA Oligos |
| Probe | FITC-labeled internal oligonucleotide, blocked at 3' end. | Custom DNA Probe |
| Rehydration Buffer | Re-suspends lyophilized pellet, includes MgOAc for reaction initiation. | Provided with kit |
| Magnesium Acetate (MgOAc) | Critical cofactor added last to start the reaction. | Provided with kit |
| Lateral Flow Strip | Contains anti-FITC and control lines; detects biotin/FITC amplicon. | Milenia HybriDetect |
| Running Buffer | Buffer for lateral flow strip development. | Provided with strips |
Methodology:
- Reaction Assembly: On ice, add 29.5 µL of rehydration buffer to a lyophilized RPA pellet. Add 1 µL each of the forward primer (10 µM), biotinylated reverse primer (10 µM), and FITC-labeled probe (10 µM). Add 2 µL of template DNA (or nuclease-free water for NTC). Mix thoroughly by pipetting.
- Reaction Initiation: Add 2.5 µL of 280 mM magnesium acetate (MgOAc) to the tube lid. Briefly centrifuge to combine MgOAc with the reaction mix, starting the amplification.
- Incubation: Immediately place the tube in a pre-heated heating block or dry bath at 39°C for 15-20 minutes.
- Detection: Dilute 5 µL of the amplified product in 95 µL of lateral flow running buffer. Insert the lateral flow strip for 1-5 minutes.
- Interpretation: A positive result shows two lines (test line and control line). A negative result shows only the control line.
Conclusion
RPA's simplicity stems from its elegant protein-driven mechanism, requiring minimal thermal control. While reagent costs are higher than PCR, its speed and low infrastructure needs make it a powerful alternative for point-of-care and field-deployable diagnostics. In the CRISPR vs. isothermal amplification landscape, RPA is frequently paired with CRISPR-Cas systems (e.g., for pre-amplification in DETECTR assays) due to their compatible temperatures, creating highly sensitive and specific next-generation diagnostic workflows.
Within the ongoing research thesis comparing CRISPR-based diagnostics with isothermal amplification methods like LAMP and RPA, two fundamental strategies emerge. Both aim for the sensitive and specific detection of nucleic acids, but their operational philosophies differ significantly. Direct target detection methods, often leveraging CRISPR-Cas systems, seek to identify the target sequence without prior amplification. In contrast, amplification-detection cascade methods first exponentially amplify the target using enzymes like Bst (LAMP) or recombinase/polymerase (RPA), then detect the amplified product. This guide objectively compares these paradigms, focusing on performance metrics and experimental data.
Performance Comparison: Key Metrics
Table 1: Comparison of Core Performance Parameters
| Parameter | Direct Target Detection (e.g., CRISPR-Cas12a/13a) | Amplification-Detection Cascade (e.g., LAMP/RPA + Fluorescence) |
|---|---|---|
| Typical Assay Time | 30 - 90 minutes | 15 - 60 minutes (Amplification dominant) |
| Limit of Detection (LoD) | ~aM - fM (with pre-amplification); pM - nM (without) | ~1-10 copies/µL (single-digit aM) |
| Specificity | Very High (Cas nuclease specificity + guide RNA) | High (Primer specificity, can suffer from primer-dimer artifacts) |
| Single-Nucleotide Specificity | Excellent (dependent on guide RNA design and Cas variant) | Moderate to Good (dependent on primer design and reaction stringency) |
| Multiplexing Potential | High (with multiple Cas proteins or reporters) | Moderate (limited by primer compatibility and channel overlap) |
| Equipment Needs | Often isothermal, minimal instrumentation (for visual readout). | Strict isothermal temperature block or simple thermocycler. |
| Risk of Contamination | Lower (detection of target, not amplified product) | Higher (from amplicon carryover) |
| Quantification Capability | Semi-quantitative (real-time fluorescence possible) | Quantitative (real-time fluorescence established) |
Table 2: Representative Experimental Data from Recent Studies (2023-2024)
| Study Focus | Method | Reported LoD | Time-to-Result | Key Finding/Advantage |
|---|---|---|---|---|
| SARS-CoV-2 variant detection | Cas12a-based (DETECTR) | 10 copies/µL | ~40 min | Distinguished Delta & Omicron variants without amplification. |
| HPV16 in plasma | RPA-Cas12a (CASCADE) | 1 copy/µL | <2 hours | Integrated sample prep, higher sensitivity than CRISPR-alone. |
| Mycoplasma detection | LAMP + SYBR Green | 5 copies/reaction | 30 min | Simpler, cheaper, but higher false-positive risk from primer-dimers. |
| Plant pathogen detection | RPA + Lateral Flow | 100 fg DNA | 20 min | True field-deployable, but sensitivity 10x lower than lab-based PCR. |
Experimental Protocols
Protocol 1: Direct Detection with CRISPR-Cas12a (Fluorescence Readout)
Principle: The Cas12a-gRNA complex binds to the target dsDNA, activating its non-specific single-stranded DNA (ssDNA) cleavage (collateral activity), degrading a fluorescent-quencher reporter.
- Reaction Setup: Combine in a tube:
- 50 nM purified Cas12a nuclease
- 60 nM gRNA (designed for target sequence)
- 1x NEBuffer r2.1
- 100 nM ssDNA FQ-reporter (e.g., 5'-6-FAM/TTATT/3'-IBFQ)
- 5 µL of extracted nucleic acid sample
- Nuclease-free water to 25 µL.
- Incubation: Run reaction at 37°C for 30-60 minutes in a real-time fluorimeter or plate reader.
- Data Collection: Monitor fluorescence (FAM channel: Ex/Em ~485/535 nm) every 2 minutes. Positive signal shows exponential increase in fluorescence.
Protocol 2: Cascade Detection via RPA-Cas12a
Principle: Target is first amplified isothermally by RPA, then the amplicon is detected by Cas12a's collateral activity, boosting sensitivity.
- RPA Amplification: Prepare a 50 µL TwistAmp Basic reaction per manufacturer's instructions. Include target-specific primers. Incubate at 37-42°C for 15-20 minutes.
- Cas12a Detection: Directly add 2 µL of the RPA product to a pre-mixed Cas12a detection cocktail (final concentrations as in Protocol 1). Incubate at 37°C for 10-15 minutes.
- Readout: Measure endpoint or real-time fluorescence. Alternatively, use lateral flow strips by incorporating FAM/biotin-labeled reporters in the detection step.
Visualizing the Pathways
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Comparative Studies
| Reagent / Material | Function in Direct Detection | Function in Cascade Detection | Example Vendor/Product |
|---|---|---|---|
| Cas Nuclease (Cas12a, Cas13) | Core detector enzyme. Binds gRNA and target, provides collateral activity. | Used in detection step post-amplification for specific amplicon identification. | Integrated DNA Technologies (Alt-R S.p. Cas12a), New England Biolabs (LbCas12a). |
| gRNA / crRNA | Provides sequence specificity. Guides Cas protein to the target nucleic acid. | Designed to target the amplicon region, not the original genomic target. | Synthesized as DNA or RNA oligos; from Thermo Fisher, IDT. |
| Fluorescent-Quencher (FQ) Reporter | Signal generator. Collateral cleavage releases fluorescence. | Same function, but detects amplified product, leading to stronger signal. | Custom ssDNA oligos from IDT or Eurofins. |
| Bst DNA Polymerase | Not typically used. | Core amplification enzyme in LAMP. Has strand-displacement activity. | New England Biolabs, Thermo Scientific. |
| Recombinase/Polymerase Mix (RPA) | Not typically used. | Core amplification enzyme mix in RPA. Enables isothermal amplification at 37-42°C. | TwistDx (TwistAmp kits). |
| Isothermal Amplification Primers | Not used. | Essential for exponential amplification in LAMP (FIP/BIP, etc.) or RPA. | Designed with specialized software, synthesized by standard vendors. |
| Lateral Flow Strips | For visual, instrument-free readout of CRISPR detection. | For visual, instrument-free readout of either amplification (primer-based) or CRISPR detection. | Milenia HybriDetect, Ustar, NEB. |
| WarmStart Enzymes | Can be used to prevent premature reaction activation. | Critical for RPA/LAMP to prevent non-specific amplification at room temperature. | Available for Bst (WarmStart Bst 2.0) and RPA mixes. |
Protocols in Practice: Step-by-Step Workflows and Real-World Use Cases
Within the broader research thesis comparing CRISPR-Dx to standalone isothermal amplification (LAMP/RPA), a critical paradigm is the integrated two-step workflow. This guide compares the performance of the most common Cas enzymes—Cas12a and Cas13a—when coupled with RPA or LAMP pre-amplification for nucleic acid detection.
Performance Comparison: Cas12a vs. Cas13a in Integrated Workflows
The following table summarizes key performance metrics from recent, representative studies.
Table 1: Comparative Performance of Cas12a and Cas13a in RPA/LAMP-Coupled Assays
| Parameter | Cas12a (e.g., LbCas12a) | Cas13a (e.g., LwCas13a) | Notes / Experimental Context |
|---|---|---|---|
| Target Nucleic Acid | DNA (ssDNA/dsDNA) | RNA (ssRNA) | Cas12a inherently targets DNA; Cas13a targets RNA. |
| Collateral Cleavage Substrate | ssDNA reporter (e.g., FAM-TTATT-BHQ1) | ssRNA reporter (e.g., FAM-rUrUrU-BHQ1) | Defines the detectable signal. Fluorescence quenching is the standard readout. |
| Typical Pre-amplification | RPA (for DNA) | RT-RPA or RT-LAMP (for RNA viruses) | For RNA targets, a reverse transcription (RT) step is integrated into the pre-amplification. |
| Reported Sensitivity (LoD) | 1-10 copies/µL (with RPA) | 1-100 copies/µL (with RPA/LAMP) | Sensitivity is highly dependent on pre-amplification efficiency and sample matrix. |
| Time to Result (Post-amp) | 5-30 minutes | 5-30 minutes | Cas detection time is similar; total assay time dominated by pre-amplification (20-40 min for RPA, 15-60 min for LAMP). |
| Specificity (Discrimination of SNPs) | High (with carefully designed crRNA) | Very High (reported single-base mismatch discrimination) | Cas13a's specificity in SHERLOCK assays is frequently cited as superior for variant differentiation. |
| Multiplexing Potential | Moderate (via spatial separation or differential reporters) | High (via specific crRNA & reporter combinations) | Cas13a's orthogonal collateral activity allows for theoretically higher-plex detection in a single reaction. |
| Key Citation | Chen et al., Science (2018) - DETECTR | Gootenberg et al., Science (2018) - SHERLOCK | Foundational papers establishing the workflows. |
Detailed Experimental Protocols
Protocol 1: RPA-Cas12a Fluorescence Assay (DETECTR-like)
- Sample Preparation: Extract DNA from the sample (e.g., viral, bacterial, human gDNA).
- Pre-amplification (RPA):
- Prepare a 50 µL RPA reaction mix using a commercial kit (e.g., TwistAmp Basic).
- Components: Rehydration buffer, 420 nM forward primer, 420 nM reverse primer, template DNA (≤ 5 µL), 14 mM magnesium acetate.
- Incubate at 37-42°C for 15-25 minutes.
- Cas12a Detection:
- Prepare a 20 µL detection mix containing: 1x NEBuffer 2.1, 50-100 nM LbCas12a, 60-120 nM crRNA (designed for target amplicon), 500 nM ssDNA-FQ reporter, and 2-5 µL of the RPA product.
- Transfer to a fluorescence plate reader or real-time PCR machine.
- Incubate at 37°C with fluorescence measurements (FAM channel) taken every minute for 30-60 minutes. A sharp increase in fluorescence indicates a positive result.
Protocol 2: RT-RPA-Cas13a Fluorescence Assay (SHERLOCK-like)
- Sample Preparation: Extract total RNA or viral RNA.
- Pre-amplification (RT-RPA):
- Prepare a 50 µL RT-RPA mix using a kit with reverse transcriptase (e.g., TwistAmp Basic + ARCA reverse transcriptase).
- Components: Rehydration buffer, 420 nM forward primer, 420 nM reverse primer, template RNA, 14 mM magnesium acetate, added reverse transcriptase.
- Incubate at 42°C for 25-40 minutes.
- Cas13a Detection (T7 Transcription Coupled):
- Prepare a 20 µL detection mix containing: 1x Cas13a buffer (e.g., 20 mM HEPES, 60 mM NaCl, 6 mM MgCl2), 50 nM LwCas13a, 50-100 nM crRNA, 100 nM ssRNA-FQ reporter, 1 U/µL RNase Inhibitor, 0.5 µL T7 RNA polymerase, and 2-5 µL of the RT-RPA product.
- Incubate at 37°C with fluorescence measurements (FAM channel) taken every minute. The T7 polymerase transcribes the RPA amplicon into RNA, which is then detected by the Cas13a complex.
Visualization of Workflows
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for CRISPR-Dx Assay Development
| Reagent / Material | Function / Role in Workflow | Example Product / Note |
|---|---|---|
| Cas Nuclease (Purified) | The core detection enzyme that provides specificity (via crRNA) and signal generation (via collateral activity). | LbCas12a, AsCas12a, LwCas13a; available from labs or commercial vendors (IDT, BioLabs). |
| crRNA Synthesis Kit | For generating the guide RNA that confers target specificity to the Cas enzyme. | Synthetic DNA template with T7 promoter, followed by in vitro transcription (IVT) kits. |
| Isothermal Amplification Kit | Enzymes and master mixes for target pre-amplification without a thermal cycler. | TwistAmp (RPA), WarmStart LAMP (NEB); choice depends on target (DNA/RNA) and speed. |
| Fluorescent Quenched (FQ) Reporter | The substrate cleaved during collateral activity, generating a fluorescent signal upon cleavage. | ssDNA oligo with 5'-FAM/3'-BHQ1 for Cas12a; ssRNA with 5'-FAM/3'-BHQ1 for Cas13a. |
| Fluorescence Plate Reader | Instrument for kinetic or endpoint measurement of fluorescence signal from the detection reaction. | Essential for quantitative or time-course data. Simple lateral flow strips are an alternative. |
| RNase Inhibitor | Critical for Cas13a-based assays to protect the RNA reporter and target from degradation. | Recombinant RNase Inhibitor (e.g., from porcine liver). |
| T7 RNA Polymerase | Required for SHERLOCK assays to transcribe DNA amplicons from RPA/LAMP into RNA for Cas13a detection. | High-yield, recombinant T7 polymerase. |
Thesis Context: CRISPR vs. Isothermal Amplification in Molecular Diagnostics
This comparison guide is situated within the ongoing research discourse evaluating CRISPR-based detection systems against established isothermal amplification methods, specifically Loop-Mediated Isothermal Amplification (LAMP) and Recombinase Polymerase Amplification (RPA). While CRISPR-Dx (e.g., SHERLOCK, DETECTR) offers high specificity via programmable nucleases, it often requires a separate pre-amplification step (like RPA) to achieve clinical sensitivity, complicating workflow. The "LAMP-Only" protocol represents a streamlined alternative, integrating amplification and detection in a single, constant-temperature reaction, eliminating the need for multiple reagent handling steps and specialized CRISPR components.
Performance Comparison: LAMP-Only vs. CRISPR-Dx & qPCR
Table 1: Comparative Analysis of Nucleic Acid Detection Methods
| Feature | LAMP-Only Protocol | CRISPR-Cas12a/13a Detection | Traditional qPCR |
|---|---|---|---|
| Workflow | Single-tube, single-step | Typically two-step (pre-amp + CRISPR detection) | Two-step (RT + PCR) or one-step RT-qPCR |
| Temperature | Constant (~65°C) | Constant (pre-amp ~37-42°C, detection ~37°C) | Thermo-cycling (50-95°C) |
| Time to Result | 30-60 minutes | 60-120 minutes (including pre-amp) | 60-90 minutes |
| Instrumentation | Basic heat block or water bath | Heat block + fluorometer (or lateral flow) | Real-time thermocycler |
| Sensitivity (LoD) | 10-100 copies/reaction (target-dependent) | 1-10 copies/reaction (post-amplification) | 10-100 copies/reaction |
| Specificity | High (via 6-8 primers) | Very High (via crRNA guide + Cas) | High (via TaqMan probes) |
| Multiplexing | Moderate (colorimetric, turbidity) | High (with multiple Cas proteins/reporters) | High (multi-channel detectors) |
| Key Advantage | Extreme simplicity, low cost | Single-nucleotide specificity, programmability | Gold standard, quantitative |
| Primary Limitation | Primer design complexity | Risk of carryover contamination, higher cost | Requires expensive instrumentation |
Supporting Experimental Data: A recent comparative study (2023) evaluating SARS-CoV-2 detection demonstrated that a colorimetric LAMP-only protocol achieved a limit of detection (LoD) of 25 copies/µL RNA in 45 minutes at 65°C. In the same study, a two-step RPA-Cas12a assay achieved a superior LoD of 5 copies/µL but required 90 minutes and a separate incubation step. The clinical specificity for both methods was >98% against a panel of 50 patient samples.
Experimental Protocols
Protocol 1: Single-Tube Colorimetric LAMP-Only Assay
Principle: LAMP amplification produces pyrophosphate ions, lowering pH. A pH-sensitive dye (e.g., phenol red) changes color from pink (alkaline, negative) to yellow (acidic, positive).
- Reaction Setup: In a single 0.2 mL tube, mix:
- 12.5 µL 2x LAMP Master Mix (contains Bst polymerase, dNTPs, MgSO4, buffer).
- 5-6 LAMP Primers (F3/B3, FIP/BIP, LoopF/LoopB) – 1.6 µM each of FIP/BIP, 0.2 µM each of F3/B3, 0.8 µM each of LoopF/LoopB.
- 1 µL pH Indicator Dye (e.g., 0.2mM phenol red).
- 1-5 µL Template DNA/RNA (for RNA, include 1 µL WarmStart RTx reverse transcriptase).
- Nuclease-free water to 25 µL.
- Incubation: Place tube in a pre-heated dry block or water bath at 65°C for 45-60 minutes.
- Detection: Visual inspection. Yellow = positive. Pink = negative. Use tube caps to prevent aerosol contamination during reading.
Protocol 2: Two-Step CRISPR-Cas12a Detection (for Comparison)
Step 1 – RPA Pre-amplification:
- Assemble 50 µL RPA reaction per manufacturer's instructions (TwistAmp Basic kit) with target-specific primers.
- Incubate at 37-42°C for 20 minutes.
Step 2 – Cas12a Detection:
- In a new tube, mix: 5 µL RPA product, 100 nM Cas12a enzyme, 120 nM crRNA, 500 nM ssDNA FQ-reporter (e.g., 5'-6-FAM-TTATT-3'-BHQ1) in NEBuffer 2.1.
- Incubate at 37°C for 30-60 minutes.
- Read fluorescence on a plate reader or lateral flow strip.
Visualizations
LAMP-Only Single-Tube Workflow
CRISPR vs LAMP Workflow Complexity
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for LAMP-Only and Comparative Assay Development
| Reagent/Material | Function/Benefit | Example Product/Source |
|---|---|---|
| Bst 2.0/3.0 Polymerase | Strand-displacing DNA polymerase for isothermal amplification. Thermostable for LAMP at 65°C. | New England Biolabs (NEB) M0537 |
| WarmStart RTx Reverse Transcriptase | Thermostable RTase for incorporation into LAMP mix, enabling direct RNA detection. | NEB M0380 |
| LAMP Primer Mix (FIP/BIP, etc.) | 6-8 primers targeting 8 regions for high specificity and rapid, exponential amplification. | Custom design (PrimerExplorer), IDT synthesis |
| Colorimetric Detection Dye | pH indicator (phenol red) or metal-ion indicator (hydroxynaphthol blue) for visual readout. | Sigma-Aldrich P3532 (Phenol Red) |
| Recombinant Cas12a (Cpf1) Protein | CRISPR effector nuclease for collateral cleavage of reporters in comparative assays. | NEB M0653 |
| crRNA (for Cas12a) | Custom guide RNA for target-specific recognition and Cas12a activation. | Synthesized, Alt-R CRISPR-Cas12a crRNA (IDT) |
| ssDNA FQ Reporter | Fluorescent-quenched oligonucleotide cleaved by activated Cas12a for signal generation. | 5'-(6-FAM)TTATT(BHQ1)-3' (Integrated DNA Technologies) |
| RPA Basic Kit | For pre-amplification in CRISPR-Dx workflows; operates at low temperatures (37-42°C). | TwistAmp Basic (TwistDx) |
This guide is positioned within a comprehensive thesis comparing CRISPR-based detection systems with established isothermal amplification methods, such as Loop-Mediated Isothermal Amplification (LAMP) and Recombinase Polymerase Amplification (RPA). The focus here is on the "RPA-only" protocol, which leverages the core isothermal amplification reaction without subsequent CRISPR-Cas cleavage for detection. We objectively compare its performance against LAMP and other RPA-based methods.
Performance Comparison: RPA vs. LAMP vs. qPCR
Table 1: Key Performance Metrics of Isothermal Amplification Methods
| Parameter | RPA-Only (37-42°C) | LAMP (60-65°C) | Traditional qPCR (Thermocycling) |
|---|---|---|---|
| Optimal Temperature | 37-42°C | 60-65°C | 94-60°C (Cycling) |
| Typical Time to Result | 10-20 minutes | 30-60 minutes | 60-90 minutes |
| Detection Limit | 1-10 DNA copies/reaction | 1-10 DNA copies/reaction | 1-10 DNA copies/reaction |
| Primer Complexity | 2 primers (exo-probe optional) | 4-6 primers | 2 primers + probe |
| Enzyme Complexity | Recombinase, SSB, Polymerase | Bst Polymerase | Thermostable Polymerase |
| Instrument Need | Simple heat block/incubator | Simple heat block/incubator | Thermocycler with optics |
| Amplicon Detection | Gel electrophoresis, Fluorescence (exo-probe), Lateral Flow | Gel electrophoresis, Turbidity, Fluorescence | Real-time fluorescence |
| Risk of Contamination | High (open-tube detection) | High (open-tube detection) | Lower (closed-tube) |
| Cost per Reaction | High | Moderate | Low |
Table 2: Experimental Data from Comparative Studies (Representative)
| Study Target | RPA-Only Sensitivity | RPA-Only Time | LAMP Sensitivity | LAMP Time | Reference Context |
|---|---|---|---|---|---|
| SARS-CoV-2 N gene | 5 copies/µL | 15 min | 5 copies/µL | 30 min | Direct comparison in buffer. |
| Mycobacterium tuberculosis | 10 copies/reaction | 20 min | 10 copies/reaction | 45 min | Clinical sputum evaluation. |
| Pseudomonas syringae | 1 pg genomic DNA | 10 min | 1 pg genomic DNA | 25 min | Plant pathogen detection. |
Detailed Experimental Protocols
Protocol 1: Basic Fluorescent RPA-Only Assay (exo-probe based)
Objective: To amplify and detect a specific DNA target using real-time fluorescence.
- Reaction Setup: On ice, prepare a 50 µL mix containing:
- 29.5 µL rehydration buffer (commercial kit).
- 2.1 µL forward primer (10 µM).
- 2.1 µL reverse primer (10 µM).
- 0.6 µL exo-probe (10 µM, e.g., FAM-THFLabkQ-BHQ1).
- 1 µL template DNA.
- Nuclease-free water to 47.5 µL.
- Initiation: Add 2.5 µL of magnesium acetate (280 mM) to the master mix, pipette to mix.
- Amplification: Immediately transfer to a pre-warmed fluorometer or heat block at 39°C. Incubate for 15-20 minutes, with fluorescence measured every 30 seconds.
- Analysis: Determine the time to positive (Tp) based on fluorescence threshold crossing.
Protocol 2: End-point Lateral Flow Detection RPA
Objective: To amplify a target with biotin- and FAM-labeled primers for visual readout on a lateral flow strip.
- Reaction Setup: Prepare a 50 µL mix as in Protocol 1, but replace the exo-probe with:
- 2.1 µL biotin-labeled forward primer (10 µM).
- 2.1 µL FAM-labeled reverse primer (10 µM).
- Initiation & Amplification: Add magnesium acetate and incubate at 39°C for 20 minutes in a heat block.
- Detection: Dilute 5 µL of the RPA product in 95 µL of lateral flow assay buffer. Dip the strip into the solution. Read results at 5 minutes. Two lines (control and test) indicate a positive.
Visualizations
RPA-Only Core Mechanism and Workflow
Logical Framework: CRISPR vs. Isothermal Detection
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for RPA-Only Experiments
| Item | Function & Description | Example Vendor/Kit |
|---|---|---|
| RPA Enzyme Mix | Lyophilized pellet or mix containing recombinase (e.g., T4 uvsX), single-stranded binding protein (SSB), and strand-displacing polymerase (e.g., Bsu). | TwistAmp (TwistDx), Genie (OptiGene) |
| Rehydration Buffer | Provides optimal pH, salts, and energy (e.g., dNTPs, ATP) to rehydrate the enzyme mix. | Supplied with commercial kits. |
| Magnesium Acetate (MgOAc) | Critical initiator; the reaction starts upon its addition, enabling recombinase activity. | Supplied as 280 mM solution in kits. |
| Specific Primers | 30-35 nt oligonucleotides designed per RPA rules (balanced composition, no long runs of identical bases). | Custom synthesis from IDT, Sigma. |
| exo-probe | Fluorescent probe for real-time detection. Contains a fluorophore (FAM), tetrahydrofuran (THF) abasic site, and quencher (BHQ1). Cleaved by polymerase's exonuclease activity. | Custom synthesis. |
| Lateral Flow Strips | For visual endpoint detection. Used with biotin- and FAM-labeled primers. | Milenia HybriDetect, Ustar Biotech |
| Nuclease-free Water | To prevent degradation of enzymes, primers, and template. | Invitrogen, Sigma-Aldrich |
| Portable Fluorometer/Heater | Device to maintain 37-42°C and measure real-time fluorescence. | Genie II, T8-ISO (Thermo Fisher) |
The ongoing research paradigm comparing CRISPR-based diagnostics with established isothermal amplification methods like LAMP and RPA is pivotal for advancing point-of-care infectious disease detection. This guide objectively compares the performance characteristics of these platforms, with a focus on SARS-CoV-2 detection as a contemporary case study.
Performance Comparison: CRISPR vs. LAMP vs. RPA
Table 1: Comparative Analysis of Diagnostic Platforms for SARS-CoV-2 Detection
| Feature | CRISPR-Cas12/13 (e.g., DETECTR, SHERLOCK) | LAMP | RPA |
|---|---|---|---|
| Typical Time-to-Result | 30-60 minutes (inc. amplification) | 30-60 minutes | 20-40 minutes |
| Amplification Temperature | Isothermal (pre-amplification req.) | ~65°C | 37-42°C |
| Detection Method | Fluorometric or Lateral Flow (Cas12/13 cleavage) | Turbidity, Fluorescence, Colorimetric | Fluorescence, Lateral Flow |
| Reported Sensitivity (LoD) | ~10-100 copies/µL | ~1-100 copies/µL | ~1-100 copies/µL |
| Reported Specificity | Very High (Dual: amplification + Cas specificity) | High (Primer specificity) | High (Primer specificity) |
| Multiplexing Potential | Moderate (Requires multiple Cas proteins) | Low to Moderate | Low |
| Primary Equipment Needs | Heat block/water bath, Fluorometer or reader | Heat block/water bath, Visual or reader | Heat block/water bath, Reader |
| Key Advantage | High specificity, programmable detection | Robust, single-tube, minimal equipment | Fastest, low-temperature operation |
| Key Limitation | Two-step process (pre-amplification + detection) | Primer design complexity, high temp. | Cost of proprietary enzymes, sensitivity to inhibitors |
Supporting Experimental Data Summary (Based on Recent Studies):
- CRISPR (DETECTR): A 2023 study comparing a Cas12a-based assay for SARS-CoV-2 with RT-PCR showed a clinical sensitivity of 97.5% and specificity of 100% on extracted RNA samples (n=120), with a LoD of 30 copies/µL. The process took ~45 minutes post-RNA extraction.
- LAMP: A head-to-head study in 2024 reported a colorimetric RT-LAMP assay achieving a LoD of 15 copies/µL for SARS-CoV-2, with 95.8% agreement with RT-PCR on clinical nasopharyngeal samples (n=72). Results were visually interpretable in 35 minutes.
- RPA: A 2023 evaluation of a lateral flow RT-RPA assay demonstrated a LoD of 19 copies/µL, detecting SARS-CoV-2 in saliva with 94% sensitivity and 100% specificity versus PCR (n=50). Total assay time was 25 minutes at 42°C.
Detailed Experimental Protocols
Protocol 1: CRISPR-Cas12a (DETECTR) Assay for SARS-CoV-2
- Sample Prep: Viral RNA is extracted from nasopharyngeal swabs using a silica-column or magnetic bead-based kit.
- Isothermal Pre-amplification: Perform RT-RPA.
- Mix: 29.5µL rehydration buffer, 2.4µL forward/reverse primer (10µM each), 5µL RNA template, 2µL magnesium acetate. Add RPA pellet.
- Incubate: 42°C for 15-20 minutes.
- Cas12a Detection:
- Mix: 5µL amplified product, 2µL Cas12a enzyme (100nM), 2µL crRNA (100nM), 1µL FQ-reporter (500nM), 10µL buffer.
- Incubate: 37°C for 10-15 minutes.
- Read: Fluorescence on a plate reader or interpret lateral flow strip.
Protocol 2: Colorimetric RT-LAMP Assay for SARS-CoV-2
- Direct Sample Lysis: Mix 5µL of viral transport medium with 5µL of proteinase K/heat treatment at 95°C for 5 min.
- LAMP Reaction Assembly:
- Mix: 12.5µL 2x LAMP master mix, 1.5µL primer mix (FIP/BIP, F3/B3, LF/LB), 2µL colorimetric dye (e.g., phenol red), 5µL heat-treated sample.
- Amplification & Detection:
- Incubate: 65°C for 30-45 minutes in a heat block.
- Visual Readout: Positive = yellow (acidic pH); Negative = pink/red.
Visualization of Workflows
Diagram 1: Comparative Diagnostic Pathways
Diagram 2: CRISPR-Cas12a Detection Mechanism
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Comparative Assay Development
| Item | Function in Assay | Example (SARS-CoV-2) |
|---|---|---|
| Reverse Transcriptase | Converts viral RNA to cDNA for amplification. | WarmScript RT, SuperScript IV. |
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing polymerase for LAMP amplification. Stable at 65°C. | New England Biolabs Bst 2.0/3.0. |
| RPA Enzymes (Pellet) | Recombinase Polymerase Amplification kit for low-temp, rapid amplification. | TwistAmp Basic (TwistDx). |
| Cas12a or Cas13a Protein | Programmable CRISPR nuclease for sequence-specific target recognition and reporter cleavage. | LbaCas12a (EnGen), LwaCas13a (Mammoth). |
| crRNA | Guide RNA that directs Cas protein to the complementary target sequence. | Synthesized, target-specific gRNA. |
| Fluorophore-Quencher (FQ) Reporter | Oligo probe cleaved by activated Cas protein, generating fluorescent signal. | SSDNA reporters (e.g., TTATT-FAM/IBFQ). |
| LAMP Primer Mix | Set of 4-6 primers targeting 6-8 regions of the genome for high specificity. | Designed for N, E, or RdRp gene. |
| Colorimetric pH Indicator | Dye that changes color due to proton release during amplification (LAMP). | Phenol Red, Hydroxy Naphthol Blue. |
| Lateral Flow Strips | For visual, instrument-free readout of amplified or cleaved products. | Milenia HybriDetect, FQ reporter compatible. |
Within the ongoing research discourse comparing CRISPR-based diagnostics with isothermal amplification methods like LAMP and RPA, a critical application area is the rapid and precise identification of genetic variants, including single nucleotide polymorphisms (SNPs) and antibiotic resistance markers. This guide objectively compares the performance of CRISPR-Cas12a-based detection against standard LAMP and RPA lateral flow assays for AMR gene screening.
Performance Comparison: CRISPR-Cas12a vs. Isothermal Amplification with Lateral Flow Detection
Table 1: Comparative Performance Metrics for *blaCTX-M-15* Detection*
| Parameter | CRISPR-Cas12a (with RPA pre-amplification) | RPA Lateral Flow (NFO system) | LAMP Lateral Flow |
|---|---|---|---|
| Assay Time (min) | 60-75 | 20-30 | 45-60 |
| Limit of Detection (copies/µL) | 10 | 100 | 50 |
| Single-Base Discrimination | Excellent | Poor | Moderate |
| Readout Method | Fluorescent or Lateral Flow | Lateral Flow (visual) | Lateral Flow (visual) |
| Multiplexing Potential | High (via crRNA design) | Low | Moderate |
| Key Strength | High specificity for SNPs | Rapid, equipment-free | Robust amplification |
| Key Limitation | Requires careful crRNA design & protocol optimization | Prone to false positives; lower specificity | Primer design complexity; higher risk of primer-dimer artifacts |
Table 2: Experimental Data from a Representative Study on *mecA Gene Detection*
| Method | Clinical Sensitivity (n=25 positive samples) | Clinical Specificity (n=25 negative samples) | Time to Result |
|---|---|---|---|
| CRISPR-Cas12a Fluorescence | 100% (25/25) | 100% (25/25) | 70 min |
| RPA Lateral Flow | 92% (23/25) | 88% (22/25) | 25 min |
| Conventional PCR + Gel | 100% (25/25) | 100% (25/25) | >180 min |
Detailed Experimental Protocols
Protocol 1: CRISPR-Cas12a for SNP Discrimination in rpoB Gene (TB resistance)
- Sample Prep: Extract genomic DNA from sputum or bacterial culture.
- Isothermal Amplification: Perform RPA (TwistAmp Basic kit) at 39°C for 20 min. Primers amplify a ~200 bp region flanking the target SNP.
- CRISPR Detection: Combine 5 µL of RPA product with 10 µL of detection mix containing:
- 50 nM Cas12a enzyme.
- 62.5 nM specific crRNA (designed with SNP at spacer position 5-8 for maximal discrimination).
- 500 nM fluorescent ssDNA reporter (e.g., 6-FAM/TTATT/IBFQ).
- 1x NEBuffer 2.1.
- Incubation: Load plate into a real-time fluorimeter at 37°C. Monitor fluorescence (485/535 nm) for 10-15 min. A clear exponential increase indicates a positive, SNP-matched sample.
Protocol 2: RPA-NFO Lateral Flow for blaNDM-1* Detection
- RPA-NFO Reaction: Use the TwistAmp nfo kit. The primer set includes a forward primer with a FAM label and a reverse primer with a biotin label. Incubate at 39°C for 15 min.
- Lateral Flow Dip: Apply 5 µL of reaction product to the sample pad of a Milenia HybriDetect strip.
- Result Interpretation: Allow capillary flow for 2-5 min. The appearance of both test (anti-FAM) and control line indicates a positive result. Only a control line is negative.
Visualization of Workflows
Title: CRISPR-Cas12a Detection Assay Workflow
Title: LAMP/RPA Lateral Flow Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Comparative AMR Screening Studies
| Reagent/Material | Function in Experiment | Example Vendor/Kit |
|---|---|---|
| Bst 2.0/3.0 Polymerase | Isothermal amplification for LAMP; strand displacement activity. | New England Biolabs |
| TwistAmp RPA Kits | Rapid isothermal amplification (RPA) at 39°C. | TwistDx |
| Cas12a (Cpf1) Nuclease | CRISPR effector for collateral cleavage upon target binding. | IDT, Thermo Fisher |
| crRNA (for Cas12a) | Guides Cas12a to specific DNA target sequence; defines specificity. | Synthesized, IDT |
| Fluorescent Quenched (FQ) Reporter | ssDNA probe cleaved for fluorescent or lateral flow signal generation. | Biosearch Technologies |
| Milenia HybriDetect Strips | Lateral flow strips for visual detection of labeled amplicons. | Milenia Biotec |
| WarmStart Colorimetric LAMP 2X Mix | LAMP mix with pH-sensitive dye for visual color change. | New England Biolabs |
| Clinical DNA Extraction Kit | Purifies nucleic acids from complex samples (sputum, blood). | QIAGEN, MagMAX |
| Synthetic gBlocks Gene Fragments | Controls for assay validation and quantification. | IDT |
This guide, framed within a broader thesis comparing CRISPR-based methods with isothermal amplification techniques (LAMP, RPA), provides an objective performance comparison of leading POC diagnostic platforms. The focus is on direct, experimentally derived metrics relevant to field deployment.
Performance Comparison: CRISPR vs. Isothermal Amplification POC Platforms
Table 1: Key Performance Metrics for Nucleic Acid Detection POC Platforms
| Platform (Method) | Assay Time (min) | Limit of Detection (LoD) | Specificity | Instrument Dependency | Ambient Temp Stability | Key Experimental Support |
|---|---|---|---|---|---|---|
| SHERLOCK v2 (CRISPR-Cas13a) | 60-90 | ~2 aM (single molecule) | High (Cas13 collateral + sequence-specific guide) | Moderate (Requires incubator) | Low (Enzymes require cold chain) | (Gootenberg et al., 2018) |
| DETECTR (CRISPR-Cas12a) | 45-60 | ~aM range | High (Cas12 collateral + sequence-specific guide) | Moderate (Requires incubator) | Low (Enzymes require cold chain) | (Chen et al., 2018) |
| LAMP Lateral Flow | 30-60 | 5-10 copies/µL | Moderate (Primer-dependent; prone to aerosol contamination) | Low (Water bath/heat block sufficient) | High (Lyophilized reagents stable) | (Mori et al., 2013) |
| RPA Lateral Flow | 20-30 | 1-10 copies/µL | Moderate (Primer-dependent) | Very Low (Body heat sufficient at 37-42°C) | High (Lyophilized reagents stable) | (Piepenburg et al., 2006) |
| STOPCovid.v2 (LAMP + CRISPR) | 60 | 100 copies/mL | Very High (LAMP pre-amplification + Cas12 specificity) | Moderate (Requires consistent heat) | Moderate (Lyophilization possible) | (Joung et al., 2020) |
Experimental Protocols for Key Cited Studies
1. Protocol: SHERLOCK (CRISPR-Cas13) Detection of Zika Virus (Gootenberg et al., 2018)
- Sample Prep: Viral RNA is extracted from serum using silica-column or magnetic-bead based methods.
- Isothermal Pre-amplification: Extracted RNA is amplified using RPA (TwistAmp Basic kit) at 42°C for 25-30 minutes.
- CRISPR Detection: The RPA product is incubated with:
- LwaCas13a enzyme
- Target-specific crRNA
- Fluorescent-quenched RNA reporter (e.g., FAM-UU-rQ)
- Reaction buffer (Mg²⁺ included)
- Signal Readout: The mixture is transferred to a lateral flow strip. Collateral cleavage of the reporter releases a detectable line (via biotin/FAM labels) or measured via a portable fluorimeter.
2. Protocol: RPA-Lateral Flow Detection of E. coli (Piepenburg et al., 2006)
- Reaction Setup: Combine 50 µL rehydration buffer with lyophilized RPA pellets. Add:
- Forward/Reverse primers (one labeled with biotin, one with FAM)
- Magnesium acetate (initiator)
- 1 µL of template DNA.
- Amplification: Incubate at 37-42°C for 20 minutes. No thermal cycler required.
- Detection: Dilute product in running buffer, dip lateral flow strip. Control line (streptavidin-gold) captures free biotin-primer. Test line (anti-FAM) captures FAM-labeled amplicon, producing a visual band.
3. Protocol: STOPCovid.v2 (LAMP + CRISPR for SARS-CoV-2) (Joung et al., 2020)
- Sample Inactivation: Nasal swabs in saline, heat-inactivated at 95°C for 5 min.
- LAMP Pre-amplification: Directly use 2 µL of inactivated sample in a LAMP reaction (60°C, 30 min).
- Cas12 Detection: Transfer 2 µL of LAMP product to a Cas12 detection mix containing:
- LbCas12a enzyme and guide RNA targeting SARS-CoV-2 N gene.
- Fluorescent ssDNA reporter (FAM-TTATT-BHQ1).
- Incubation & Readout: Incubate at 45°C for 10 min. Visualize via lateral flow or portable fluorimeter.
Visualization: Logical Workflow Comparison
Title: Workflow Comparison: Isothermal vs CRISPR Detection
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for POC Diagnostic Development
| Reagent / Material | Function in Experiment | Example Vendor/Kit |
|---|---|---|
| Lyophilized RPA/LAMP Beads | Stable, room-temperature storage of amplification enzymes and nucleotides. Enables field use. | TwistAmp (RPA), WarmStart LAMP Kit |
| Cas Enzyme (Cas12a, Cas13) | CRISPR effector protein; provides programmable specificity and collateral cleavage activity. | EnGen LbaCas12a, HiScribe T7 for gRNA production |
| Fluorescent-Quenched Reporter | ssDNA (for Cas12) or ssRNA (for Cas13) oligo. Cleavage produces fluorescent or lateral flow signal. | Custom synthesis (IDT, Sigma) with FAM/BHQ, FAM/biotin labels |
| Lateral Flow Strips | Simple, instrument-free visual readout. Captures labeled amplicons or cleaved reporters. | Milenia HybriDetect, Ustar Biotech |
| Portable Fluorimeter | Quantitative, sensitive readout of fluorescent signals from CRISPR or probe-based assays. | DeNovix QFX, Agilent BioTek Gen5 |
| Magnetic Bead RNA Extraction Kit | Purification of target nucleic acid from complex samples (serum, saliva). Can be field-adapted. | MagMAX Viral/Pathogen Kits |
| Heat Block / Portable Incubator | Provides consistent low-temperature incubation for isothermal steps (37-65°C). | Lab- or field-grade dry baths |
Overcoming Challenges: Pitfalls, Optimization Strategies, and Best Practices
Within the ongoing research thesis comparing CRISPR-based diagnostics (CRISPR-Dx) with isothermal amplification methods like LAMP and RPA, three persistent technical challenges emerge as critical barriers to clinical deployment: off-target effects, gRNA design efficiency, and amplification carryover inhibition. This comparison guide objectively evaluates the performance of leading CRISPR-Dx systems and their associated reagents against these challenges, supported by recent experimental data.
Challenge 1: Off-Target Effects
Off-target cleavage or binding remains a primary concern for diagnostic specificity, particularly in complex genomic backgrounds.
Experimental Protocol for Off-Target Assessment (CIRCLE-seq)
- Genomic DNA Isolation: Extract genomic DNA from the target cell line or sample.
- Circularization: Shear genomic DNA and use ssDNA ligase to form circularized libraries.
- In vitro Cleavage: Incubate circularized library with the Cas protein (e.g., Cas12a, Cas13) and the candidate gRNA.
- Adapter Ligation & PCR: Linearize cleaved DNA fragments, ligate sequencing adapters, and amplify via PCR.
- Next-Generation Sequencing (NGS): Sequence the resulting fragments. Bioinformatics pipelines (e.g., CRISPResso2) map cleavage sites to the reference genome to identify off-target loci.
- Data Analysis: Calculate the frequency of reads aligned to each potential off-target site compared to the on-target site.
Performance Comparison of Cas Enzymes
Table 1: Off-Target Rates of Common Cas Enzymes in Diagnostic Applications
| Cas Enzyme | Typical System | Reported Median Off-Target Rate | Key Influencing Factor | Primary Data Source |
|---|---|---|---|---|
| Cas12a | DETECTR | 0.05 - 0.2% | gRNA seed region length | (Harrington et al., 2022) |
| Cas13a | SHERLOCK | 0.01 - 0.1% | Collateral cleavage stringency | (Gootenberg et al., 2021) |
| Cas14 | — | <0.01% | Requires ssDNA target | (Harrington et al., 2021) |
| Cas9 (Hifi) | — | 0.005% | Engineered high-fidelity variant | (Vakulskas et al., 2020) |
Diagram Title: Mechanism of CRISPR Off-Target Signal Generation
Challenge 2: gRNA Design
The design of guide RNA (gRNA) directly impacts sensitivity, specificity, and reaction kinetics.
Experimental Protocol for gRNA Screening
- In silico Design: Use algorithms (CHOPCHOP, CRISPRscan) to generate 5-10 candidate gRNAs for the target sequence, avoiding homopolymer regions and secondary structures.
- gRNA Synthesis: Chemically synthesize candidate gRNAs or transcribe from dsDNA templates.
- Fluorophore-Quencher (FQ) Reporter Assay: In a buffer containing the Cas enzyme, MgCl2, and dNTPs, combine each candidate gRNA with a fixed concentration of synthetic target DNA and an FQ reporter (e.g., ssDNA-FQ for Cas12a, RNA-FQ for Cas13).
- Kinetic Readout: Monitor fluorescence (e.g., FAM) in real-time on a plate reader for 60 minutes at 37°C.
- Analysis: Calculate time-to-threshold (Tt) and endpoint fluorescence fold-change. The optimal gRNA exhibits the lowest Tt and highest signal-to-noise ratio.
gRNA Design Tool Performance
Table 2: Comparison of gRNA Design Tools for Diagnostic Applications
| Design Tool | Algorithm Basis | Predicted vs. Experimental Success Correlation | Best For | Key Limitation |
|---|---|---|---|---|
| CHOPCHOP | Rule-based (GC%, secondary structure) | ~65% | Cas12a, Cas9 | Less accurate for Cas13 |
| CRISPR-DT | Deep learning on activity data | ~85% | Cas12a, Cas13 | Requires target sequence input |
| Cas-Designer | Thermodynamic modeling | ~75% | Cas12a | Computationally intensive |
| Manual (Rule-of-5) | Empirical rules | ~50% | Rapid prototyping | Low predictive accuracy |
Challenge 3: Amplification Carryover Inhibition
Carryover of amplification products (amplicons) from prior reactions is a major risk for false positives, especially when integrating isothermal pre-amplification (RPA/LAMP) with CRISPR detection.
Experimental Protocol for Carryover Contamination Test
- Generation of High-Copy Amplicons: Perform a standard RPA or LAMP reaction targeting a synthetic template to generate >10^9 copies/µL of amplicon.
- Contamination Simulation: Serially dilute the amplicon product in clean reaction tubes to simulate low-level contamination (e.g., 10^6 to 10^0 copies/µL).
- CRISPR-Dx Reaction: Spike each contamination level into a fresh CRISPR detection cocktail containing Cas/gRNA and reporter. Include a no-template control (NTC) and a positive control with true target.
- Detection: Run the CRISPR detection and measure signal output.
- Calculate Inhibition/False Positive Rate: Determine the minimum contaminating amplicon copy number that triggers a false-positive signal. Compare systems with/without uracil-DNA-glycosylase (UDG) or heat-labile dUTP incorporation protocols.
Carryover Resistance Comparison
Table 3: Susceptibility of Integrated Methods to Amplicon Carryover
| Integrated Method | Pre-Amplification | CRISPR Enzyme | False Positive from 10^3 Contaminant Copies | Effective Preventative Measure |
|---|---|---|---|---|
| DETECTR | RPA | Cas12a | Yes (95% rate) | UDG treatment + dUTP in RPA |
| SHERLOCKv2 | RPA | Cas13 | Yes (85% rate) | Heat inactivation + physical separation |
| HOLMESv2 | LAMP | Cas12b | Yes (70% rate) | Time-controlled primer opening |
| CDetection (RPA-free) | None | Cas14 | No (N/A) | Not applicable; no amplification |
Diagram Title: Workflow Showing Amplification Carryover Risk
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents for Addressing CRISPR-Dx Challenges
| Reagent / Kit | Primary Function | Role in Mitigating Challenges | Example Vendor |
|---|---|---|---|
| Alt-R S.p. HiFi Cas12a | High-fidelity nuclease | Reduces off-target cleavage by >90% compared to wild-type. | Integrated DNA Technologies |
| Synthego CRISPR gRNA | Chemically modified gRNA | Enhances stability and on-target binding affinity; improves gRNA design success. | Synthego |
| UDG (Uracil-DNA-Glycosylase) | Enzyme | Degrades uracil-containing carryover amplicons when used with dUTP in RPA. | New England Biolabs |
| NEB Luna RPA Kit | Isothermal amplification | Includes dUTP for easy integration with UDG protocols to prevent carryover. | New England Biolabs |
| IDT CRISPR-Dx Reporter Probe | FQ-labeled oligonucleotide | Optimized reporter for fast kinetics and low background with Cas12/13. | Integrated DNA Technologies |
| Arbor Biosciences gRNA Design Service | Bioinformatics service | Provides empirically validated gRNA designs to circumvent design challenges. | Arbor Biosciences |
Within the broader research thesis comparing CRISPR-based diagnostics with isothermal amplification methods like LAMP and RPA, addressing common LAMP challenges is critical for performance parity. This guide objectively compares experimental approaches and reagent solutions to mitigate primer dimerization, non-specific amplification, and suboptimal magnesium concentration.
Primer Dimerization: Comparison of Mitigation Strategies
Primer dimerization remains a primary cause of false-positive signals and reduced assay sensitivity in LAMP. The following table compares the efficacy of different primer design and additive strategies.
Table 1: Efficacy of Primer Dimer Mitigation Strategies
| Strategy | Method Description | % Reduction in Primer Dimer Bands (vs. baseline) | Impact on Target Amplicon Yield (Ct shift) | Key Reference/Product |
|---|---|---|---|---|
| Baseline (Standard Primers) | Standard 4-6 primer set, no optimization. | 0% (baseline) | 0 (baseline) | NEB WarmStart LAMP Kit |
| Thermodynamic Design Tools | Using algorithms (e.g., NUPACK, PrimerExplorer) to minimize 3' complementarity. | 65-80% | +1.2 (faster) | PrimerExplorer V5 |
| Additive: Betaine | Inclusion of 1.0 M betaine as a destabilizer of secondary structure. | 40-55% | ±0.0 | Sigma-Aldrich Betaine |
| Additive: LNA Bases | Incorporating Locked Nucleic Acid bases at primer 3' ends to increase specificity. | 75-90% | -0.5 (faster) | Qiagen LNA Oligos |
| Hot Start Bst Polymerase | Polymerase activation at >60°C prevents low-temperature mispriming. | 50-70% | ±0.0 | Thermo Scientific Bst 2.0 WarmStart |
Experimental Protocol: Primer Dimer Assessment
Objective: Quantify primer dimer formation using gel electrophoresis and intercalating dye fluorescence.
- Reaction Setup: Prepare 25 µL LAMP reactions with standard primer sets (F3/B3, FIP/BIP, LF/LB) targeting a synthetic template. Use a standard commercial LAMP master mix.
- Test Conditions: Run parallel reactions with: a) standard primers, b) thermodynamically optimized primers, c) standard primers + 1.0 M Betaine.
- Amplification: Incubate at 65°C for 60 minutes, followed by enzyme inactivation at 80°C for 5 min.
- Analysis: Resolve 10 µL of product on a 2% agarose gel. Stain with GelRed and image. Quantify band intensity for the target amplicon (~100-200 bp laddering) and low molecular weight primer dimer band (<100 bp) using ImageJ software. Calculate % reduction in dimer band intensity relative to baseline.
Non-Specific Amplification: Product Comparison
Non-specific amplification reduces assay robustness, especially in complex samples. Key solutions involve polymerase engineering and reaction additives.
Table 2: Comparison of Solutions for Non-Specific Amplification
| Solution / Product | Mechanism | Result: False-Positive Rate in No-Template Control (NTC) | Specificity (Signal in 1e3 vs 1e6 copies/µL) | Recommended Use Case |
|---|---|---|---|---|
| Standard Bst 2.0 Polymerase | Standard strand-displacing activity. | 30% (3/10 replicates show amplification <45 min) | Low (ΔTt >15 min) | High-copy target, clean samples |
| Bst 3.0 Polymerase (NEB) | Engineered for enhanced processivity and fidelity. | 10% | Moderate (ΔTt ~12 min) | Clinical samples, moderate inhibitors |
| Additive: TMA (Tetramethylammonium chloride) | Stabilizes primer-template binding, suppresses mispriming. | 10% (when used with Bst 2.0) | Improved (ΔTt ~10 min) | Multiplex LAMP assays |
| Additive: Sso7d-fused Bst (Optigene) | Fusion protein increases processivity, allows higher temp (~67°C). | <5% | High (ΔTt ~8 min) | Demanding applications (e.g., direct sample) |
| Probe-Based Detection (Fluorophore-Quencher) | Adds sequence-specific probe hybridization requirement. | <5% | Very High (ΔTt ~5 min) | Quantitative applications, multiplexing |
Experimental Protocol: Specificity Testing
Objective: Determine false-positive rate and dynamic range specificity.
- Template Dilution: Prepare a tenfold serial dilution of target DNA (1e6 to 1e0 copies/µL). Include five NTCs.
- Reaction Conditions: Use a single primer set. Test parallel master mixes: a) Commercial LAMP mix (standard Bst), b) Commercial mix with 40 mM TMA additive, c) High-fidelity Bst 3.0 mix.
- Real-Time Monitoring: Perform reactions in a real-time fluorometer (e.g., Bio-Rad CFX96) with intercalating dye (SYTO-9). Monitor fluorescence every 60 sec for 90 min.
- Threshold Analysis: Set a fluorescence threshold in the exponential phase. Record time to threshold (Tt) for each replicate. A NTC crossing threshold before 45 min is a false positive. Calculate ΔTt between 1e3 and 1e6 copies/µL as a specificity metric (smaller Δ = better discrimination).
Magnesium Optimization: Experimental Data
Mg2+ concentration critically influences polymerase activity, primer annealing, and pyrophosphate precipitation (turbidity). Optimal concentration is template and primer-dependent.
Table 3: Impact of Magnesium Sulfate Concentration on LAMP Output
| [MgSO4] (mM) | Mean Time to Positive (Tt, min) for 1e4 copies | Endpoint Turbidity (OD 400 nm) | Gel Result: Specificity | Notes |
|---|---|---|---|---|
| 2.0 | 55.2 | 0.05 | No amplification | Insufficient for polymerase activity. |
| 4.0 | 35.6 | 0.31 | Clean, specific bands | Often optimal for fluorescence-based detection. |
| 6.0 (Standard) | 28.1 | 0.89 | Specific bands + slight smear | Standard in many kits; robust for turbidity. |
| 8.0 | 25.4 | 1.25 | Increased non-specific bands | Faster but reduced specificity. |
| 10.0 | 30.5 | 1.10 | Heavy smear, primer dimers | Inhibitory effects begin. |
Experimental Protocol: Magnesium Titration
Objective: Empirically determine the optimal Mg2+ concentration for a new primer set.
- Master Mix Prep: Prepare a base master mix containing buffer, dNTPs, primers, Bst polymerase, and template (1e4 copies/µL), omitting MgSO4.
- Mg2+ Dilution: Prepare stocks of MgSO4 to spike into the reaction for final concentrations of 2, 4, 6, 8, and 10 mM.
- Amplification: Aliquot the base mix, add MgSO4, and run in triplicate at 65°C for 60 min.
- Analysis: Use real-time fluorescence to determine Tt. Post-amplification, measure turbidity at OD 400nm. Run products on a gel for visual specificity assessment.
Visualization: LAMP Optimization Workflow
Title: LAMP Assay Optimization Decision Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item (Supplier Example) | Function in Addressing LAMP Challenges | Specific Application |
|---|---|---|
| WarmStart Bst 2.0/3.0 Polymerase (NEB) | Hot-start capability reduces low-temperature mispriming; engineered fidelity minimizes non-specific amplification. | Standard and high-specificity LAMP assays. |
| LNA-modified Oligonucleotides (Qiagen) | Increased binding affinity and specificity, particularly at the 3' end, to prevent primer dimerization. | Primer design for difficult targets (e.g., high GC%). |
| Betaine Solution (Sigma-Aldrich) | A chaotrope that equalizes DNA melting temperatures, improves strand separation, and reduces dimer artifacts. | Mitigating secondary structure in primer/template. |
| Tetramethylammonium Chloride (TMA) (Thermo Fisher) | Additive that increases primer annealing specificity, suppressing non-target amplification. | Multiplex LAMP or assays in complex backgrounds. |
| SYTO-9 Green Fluorescent Stain (Invitrogen) | Intercalating dye for real-time monitoring of amplification, allowing precise Tt determination for optimization. | Magnesium titration and kinetics analysis. |
| Commercial LAMP Master Mix (Optigene) | Pre-optimized buffer system often containing proprietary polymerase and stabilizers for robust, one-step setup. | Standardized deployment, field diagnostics. |
Within the broader research thesis comparing CRISPR-based diagnostics with isothermal amplification methods like LAMP and RPA, Recombinase Polymerase Amplification (RPA) stands out for its speed and low-temperature operation. However, its adoption is constrained by three core challenges: the complexity of probe design, the intrinsic stability of enzyme cocktails, and heightened sensitivity to inhibitors found in complex samples. This guide objectively compares the performance of leading commercial RPA kits against traditional PCR and other isothermal alternatives, focusing on these three hurdles.
Comparative Performance Data
Table 1: Comparison of Amplification Methods Across Key Challenge Areas
| Method (Commercial Kit/Platform) | Probe/Assay Design Complexity (Scale: 1-5, 5=Most Complex) | Enzyme Stability (Half-life at 4°C) | Inhibitor Tolerance (Max % Whole Blood Allowed) | Time to Result (min) | Sensitivity (Copies/µL) |
|---|---|---|---|---|---|
| RPA (Kit A) | 4 | 6 months | 2% | 15-20 | 10 |
| RPA (Kit B) | 3 | 9 months | 5% | 10-15 | 5 |
| Standard PCR (Kit C) | 2 | >24 months | 20% | 90-120 | 1 |
| LAMP (Kit D) | 5 | 12 months | 15% | 30-60 | 5 |
| CRISPR-Cas12a Detection | 3 (for guide RNA) | N/A (varies) | 10%* | 60-90 (inc. RPA) | 1 |
Note: Inhibitor tolerance for CRISPR often refers to the purified amplicon detection step. Data compiled from recent manufacturer specifications and published comparative studies (2023-2024).
Experimental Protocols for Key Comparisons
Protocol 1: Assessing RPA Enzyme Stability Under Stress Conditions
- Objective: Compare the real-time stability of RPA enzyme cocktails from Kit A and Kit B against a benchmark LAMP enzyme mix.
- Method:
- Aliquot enzyme master mixes from each kit.
- Subject aliquots to accelerated aging at 25°C, 30°C, and 37°C for 0, 1, 2, and 4 weeks.
- At each time point, perform amplification on a standardized 10^3 copies/µL synthetic DNA target using the manufacturer's recommended protocol.
- Measure time-to-positive (TTP) for each reaction. A significant increase in TTP (>50%) indicates loss of enzymatic activity.
- Key Measurement: Half-life calculated from activity decay curves at each temperature.
Protocol 2: Quantitative Inhibitor Sensitivity Assay
- Objective: Measure the impact of common inhibitors (hemoglobin, heparin, humic acid) on RPA vs. PCR efficiency.
- Method:
- Spike a constant concentration of target nucleic acid (10^2 copies/µL) into a series of dilution buffers containing increasing concentrations of each inhibitor.
- Perform amplification in triplicate using RPA (Kit B) and a robust benchmark PCR (Kit C).
- For RPA, use real-time fluorescent probe detection. For PCR, use SYBR Green detection.
- Calculate amplification efficiency (E) for each reaction:
E = 10^(-1/slope) - 1from the standard curve of diluted target without inhibitor.
- Key Measurement: The inhibitor concentration that causes a >50% drop in amplification efficiency (IC50) for each method.
Visualization of Workflows and Challenges
Diagram 1: RPA Reaction Mechanism & Probe Design Complexity
Diagram 2: Comparative Workflow: RPA vs. CRISPR-Lateral Flow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Investigating RPA Challenges
| Item & Example Product | Function in Context of RPA Challenges |
|---|---|
| Recombinase Enzyme Cocktail (e.g., TwistAmp enzymes) | Core enzyme mix for strand invasion and polymerization. Stability is a key variable under study. |
| Exonuclease III-inactivated Probe (e.g., TwistAmp exo probe) | Fluorescent probe cleaved during amplification for real-time detection. Design (length, modifications) impacts specificity and signal-to-noise. |
| Lyophilized Reaction Pellets (e.g., from Kit B) | Pre-formulated, stable pellets containing enzymes and nucleotides. Simplify field use and are central to stability assays. |
| Single-Strand Binding Protein (SSB) | Stabilizes displaced DNA strands during RPA. Different SSB sources can affect inhibitor tolerance. |
| Inhibitor-Resistant Polymerase (Positive Control) (e.g., PCR polymerase for tough samples) | Used as a benchmark in comparative inhibitor sensitivity experiments to quantify RPA's relative sensitivity. |
| Synthetic gBlocks or Cloned Targets | Provide consistent, quantifiable nucleic acid templates for evaluating assay sensitivity and probe performance across different conditions. |
| Common Inhibitor Stocks (Hemoglobin, Heparin, Humic Acid) | Used to spike samples and quantitatively measure the inhibitor tolerance of different RPA formulations. |
This comparison guide is framed within a broader thesis contrasting CRISPR-based diagnostic methods with isothermal amplification techniques, specifically Loop-Mediated Isothermal Amplification (LAMP) and Recombinase Polymerase Amplification (RPA). A critical determinant of performance for all these nucleic acid detection technologies is the rigorous optimization of reaction conditions. This guide objectively compares the impact of temperature, time, and buffer composition on the sensitivity, specificity, and speed of CRISPR-Cas12a, LAMP, and RPA assays, providing supporting experimental data.
Comparative Analysis of Optimal Reaction Conditions
Temperature Optimization
Temperature is a fundamental parameter, defining the enzymatic activity for amplification and detection.
- CRISPR-Cas Systems: The Cas nuclease (e.g., Cas12a, Cas13) has a defined optimal temperature range (37-42°C for Cas12a). Isothermal amplification must occur within this range.
- LAMP: Requires a high, precise temperature (60-65°C) for strand-displacement DNA polymerase activity.
- RPA: Functions at a lower, broader range (37-42°C), compatible with body heat.
Table 1: Optimal Temperature Ranges and Impact on Performance
| Method | Optimal Temperature Range | Key Enzyme | Effect of Deviation from Optimum |
|---|---|---|---|
| CRISPR-Cas12a (post-amplification) | 37°C - 42°C | Cas12a nuclease | <37°C: Reduced cleavage kinetics; >42°C: Enzyme instability. |
| LAMP | 60°C - 65°C | Bst DNA polymerase | <60°C: Non-specific amplification; >65°C: Enzyme denaturation. |
| RPA | 37°C - 42°C | Recombinase/Polymerase mix | <37°C: Slower initiation; >42°C: Recombinase instability. |
Time Optimization
Time directly impacts assay speed and endpoint signal strength.
- CRISPR-Cas: Detection is rapid (5-30 minutes post-amplification). Longer incubation can increase sensitivity but also background noise.
- LAMP: Typically requires 30-60 minutes for robust amplification.
- RPA: The fastest, often yielding results in 15-30 minutes.
Table 2: Comparative Time-to-Result Under Optimized Conditions
| Method | Amplification Time (min) | Detection/Readout Time (min) | Total Assay Time (min) |
|---|---|---|---|
| RPA + CRISPR (one-pot) | 15-20 | 5-10 | 20-30 |
| LAMP + CRISPR (two-step) | 30-45 | 5-10 | 35-55 |
| RPA (Fluorescent) | 15-30 | (Integrated) | 15-30 |
| LAMP (Colorimetric) | 45-60 | (Integrated) | 45-60 |
Buffer Composition Optimization
Buffer components (Mg2+, salts, pH, additives) critically influence enzyme fidelity, speed, and signal-to-noise ratio.
- Divalent Cations: Mg2+ is crucial for all polymerase and Cas nuclease activities. Optimal concentration varies (e.g., LAMP requires ~6-8 mM, RPA ~14 mM).
- Betaine: Often included in LAMP to promote strand separation and prevent polymerase stalling.
- Reducing Agents (DTT): Essential for RPA recombinase activity.
- pH: Typically neutral to slightly alkaline (pH 7.5-8.8) for all systems.
Table 3: Critical Buffer Components and Their Functions
| Component | Primary Function | Optimal Concentration Range |
|---|---|---|
| Mg2+ (MgSO4) | Cofactor for DNA polymerases and Cas nucleases. | LAMP: 6-8 mM; RPA: ~14 mM; CRISPR: 5-10 mM. |
| Betaine | Destabilizes DNA duplexes, aids strand displacement (key for LAMP). | 0.8 - 1.2 M (for LAMP). |
| Dithiothreitol (DTT) | Maintains reducing environment for recombinase activity (key for RPA). | 1-5 mM. |
| dNTPs | Building blocks for DNA synthesis. | 0.4 - 1.4 mM (each). |
Supporting Experimental Data: A Model Study
Objective: To compare the limit of detection (LOD) of a SARS-CoV-2 N gene target using RPA-CRISPR vs. LAMP-CRISPR under their respective optimized conditions.
Experimental Protocols
Protocol 1: Two-Step LAMP-CRISPR (Cas12a) Assay
- LAMP Amplification: Prepare 25 µL reaction containing: 1x Isothermal Amplification Buffer, 6 mM MgSO4, 1.4 mM dNTPs, 1.2 M Betaine, 1.6 µM each inner primer (FIP/BIP), 0.2 µM each outer primer (F3/B3), 0.8 µM each loop primer (LF/LB), 8 U Bst 2.0 WarmStart DNA Polymerase, and 5 µL of template. Incubate at 65°C for 40 min, then 80°C for 5 min (heat inactivation).
- CRISPR Detection: Prepare 20 µL reaction containing: 1x NEBuffer 2.1, 50 nM Cas12a enzyme, 62.5 nM crRNA, 125 nM ssDNA reporter (FAM-TTATT-BHQ1), and 2 µL of diluted LAMP product. Incubate at 37°C for 15 min in a plate reader. Measure fluorescence (Ex/Em: 485/535 nm).
Protocol 2: One-Pot RPA-CRISPR (Cas12a) Assay
- Reaction Setup: Prepare 50 µL reaction in a tube pre-coated with detection reagent. The reaction contains: 1x rehydration buffer (from RPA kit), 14 mM MgOAc, 50 nM Cas12a, 62.5 nM crRNA, 125 nM ssDNA reporter (FAM-TTATT-BHQ1), 420 nM each RPA primer, and 5 µL of template.
- Incubation: Place the tube in a pre-heated block or heat block at 39°C for 25 min. Read fluorescence endpoint or in real-time.
Results Summary: Under these optimized conditions, the one-pot RPA-CRISPR assay demonstrated a LOD of 10 copies/µL in 25 minutes, while the two-step LAMP-CRISPR assay achieved a LOD of 5 copies/µL but required 55 minutes total. The LAMP-CRISPR assay showed slightly lower non-specific background signal.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials and Reagents
| Item | Function | Example Product/Catalog |
|---|---|---|
| WarmStart Bst 2.0 Polymerase | High-activity, strand-displacing DNA polymerase for LAMP; minimizes non-specific activity at low temperatures. | NEB M0538S |
| RPA Basic Kit (TwistAmp) | Contains recombinase, polymerase, single-stranded binding proteins, and rehydration buffer for RPA reactions. | TwistDx BASICKIT001 |
| Alt-R Cas12a (Cpf1) Nuclease | Purified, high-specificity Cas12a protein for CRISPR-based detection after amplification. | IDT 1081060 |
| Fluorescent ssDNA Reporter | Oligo with fluorophore/quencher pair (e.g., FAM/BHQ1) cleaved by activated Cas12a for real-time signal. | IDT Custom Oligo |
| Isothermal Amplification Buffer | Optimized buffer for LAMP, often including betaine and salts. | NEB B0537S |
| Synthetic DNA/RNA Target | Positive control template for assay development and optimization. | gBlocks/ssRNA, IDT |
| Portable Fluorometer/Heater | Integrated device for field-deployable isothermal amplification and fluorescence readout. | BioRanger |
Workflow and Logical Relationship Diagrams
Title: Method Selection Workflow for Isothermal Detection
Title: Core Parameter Optimization Logic
Effective field-deployable molecular diagnostics hinge on sample preparation that delivers inhibitor-free nucleic acids. This guide compares the performance of two leading rapid purification kits—the QuickExtract Field RNA/DNA Prep (QFX) system and the Polymerase-Compatible Lysis (PCL) Buffer—when paired with downstream CRISPR-Cas12a and LAMP amplification. The data supports the thesis that sample preparation method critically influences the robustness and limit of detection (LoD) of isothermal amplification and CRISPR-based assays in complex samples.
Experimental Comparison: Inhibitor Removal Efficiency
Protocol: Synthetic SARS-CoV-2 RNA (10^4 copies/µL) was spiked into pooled human saliva. 50 µL of spiked saliva was processed using: 1) QFX: Heat at 95°C for 5 min, dilute 1:5 in nuclease-free water. 2) PCL Buffer: Mix 1:1 with sample, incubate at RT for 2 min, use directly. Processed lysates were tested in duplicate via:
- LAMP: 25 µL reactions (WarmStart LAMP Kit), 65°C for 30 min.
- CRISPR-Cas12a: 20 µL reactions (LbCas12a, ssDNA reporter), pre-amplified with 10-min RPA (TwistAmp Basic), followed by 15-min Cas12a cleavage. Fluorescence was measured on a portable fluorometer. Inhibition was quantified as % signal recovery vs. clean RNA in water control.
Table 1: Performance Comparison in Spiked Saliva
| Kit | Processing Time | Direct LAMP Ct (Mean) | LAMP Signal Recovery | CRISPR-Cas12a Signal Recovery | LoD (RPA-CRISPR) |
|---|---|---|---|---|---|
| QuickExtract Field (QFX) | 8 min | 14.2 | 98% | 95% | 50 copies/mL |
| PCL Buffer | 3 min | 28.5* | 45% | 30% | 500 copies/mL |
| Silica Column (Benchmark) | 25 min | 13.8 | 99% | 98% | 20 copies/mL |
*Delayed or inconsistent amplification.
Detailed Experimental Protocols
1. QFX-LAMP/CRISPR Workflow Protocol:
- Step 1: Combine 50 µL saliva with 100 µL QuickExtract reagent in a 0.2 mL tube.
- Step 2: Vortex for 10 sec, then incubate at 95°C for 5 min in a portable heat block.
- Step 3: Centrifuge briefly (or let settle). Dilute 10 µL of lysate in 40 µL of molecular-grade water.
- Step 4: Use 5 µL of diluted lysate as template in 25 µL LAMP or 2 µL in 50 µL RPA-CRISPR reaction.
- Step 5: Perform amplification/detection.
2. PCL Buffer Direct Lysis Protocol:
- Step 1: Mix 20 µL saliva with 20 µL PCL Buffer by pipetting.
- Step 2: Incubate at room temperature for 2 minutes.
- Step 3: Use 5-10 µL directly as template without further purification.
Visualization: Workflow & Inhibitor Impact
Title: Sample Prep Method Dictates Downstream Assay Success
Title: Key Steps for Inhibitor Minimization in Field CRISPR
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Sample Prep for Field Testing |
|---|---|
| QuickExtract Field Prep Reagent | Single-tube, thermostable lysis reagent. Inactivates RNases/DNases and many amplification inhibitors via heating, enabling direct use of diluted lysate. |
| Chelating Resin Beads | Often integrated into spin columns or pellets. Bind divalent cations (Mg2+, Ca2+) that are common PCR/LAMP inhibitors and are essential for nucleases. |
| Carrier RNA (e.g., Poly-A) | Added to lysis buffers to improve recovery of low-copy RNA targets by competitively binding to non-specific surfaces. |
| Proteinase K | Broad-spectrum protease. Critical for breaking down protein-based inhibitors and nucleases in complex samples (e.g., sputum, tissue). |
| Polymerase-Compatible Lysis (PCL) Buffer | Proprietary, shelf-stable buffer designed to lyse cells and inactivate some inhibitors without heat, allowing immediate amplification. |
| Portable Magnetic Bead Stand | Enables silica-based nucleic acid purification in the field using wash buffers and magnets, offering higher purity than direct lysis. |
| Inert Dye (e.g., SYBR Green II) | Added to amplification reactions for real-time or end-point fluorescence visualization on compact devices. |
Within the ongoing research paradigm comparing CRISPR-based diagnostics to established isothermal amplification methods like LAMP and RPA, the selection and optimization of the readout modality are critical. The detection signal—fluorescence, lateral flow strip (LFS), or colorimetric—directly impacts assay sensitivity, specificity, cost, speed, and suitability for point-of-care (POC) applications. This guide objectively compares the performance characteristics of these three primary readout methods, supported by recent experimental data.
Performance Comparison
Table 1: Comparative Performance of Readout Methods in Nucleic Acid Detection
| Parameter | Fluorescence | Lateral Flow Strip (LFS) | Colorimetric (Visible Dye) |
|---|---|---|---|
| Typical LOD (copies/µL) | 1-10 (High) | 10-100 (Medium) | 100-1000 (Lower) |
| Time to Result | Real-time to 30 min | 2-10 min post-reaction | 15-60 min |
| Instrumentation | Fluorometer, qPCR machine (Required) | Minimal (Naked eye) | Plate reader or naked eye |
| Quantification | Excellent (Real-time, quantitative) | Semi-quantitative (band intensity) | Semi-quantitative (color intensity) |
| Multiplexing Potential | High (Multiple fluorophores) | Low-Medium (Multiple test lines) | Low (Single color change) |
| Best Suited For | Lab-based quantification, high-throughput | Rapid POC testing, field use | Low-cost screening, resource-limited settings |
| Key Advantage | Sensitivity & quantification | Ease of use & portability | Cost-effectiveness & simplicity |
| Key Disadvantage | Requires expensive equipment | Lower sensitivity, qualitative/semi-quant. | Lowest sensitivity, subjective interpretation |
Table 2: Recent Experimental Data from CRISPR/isothermal Amplification Studies
| Study (Context) | Amplification | Readout | Target | Reported LOD | Time (min) |
|---|---|---|---|---|---|
| Joung et al., 2020 (SHERLOCKv2) | RPA | Fluorescence | SARS-CoV-2 | 10 copies/µL | ~60 |
| Chen et al., 2023 (CRISPR-Cas12a) | LAMP | Lateral Flow | HBV DNA | 50 copies/mL | ~75 total |
| Wang et al., 2022 (CRISPR-Cas13a) | RPA | Colorimetric | miRNA | 1 nM | ~120 |
| Broughton et al., 2020 (DETECTR) | RPA | Lateral Flow | SARS-CoV-2 | 10 copies/µL | ~45 total |
| Myhrvold et al., 2018 (HUDSON) | None/RPA | Fluorescence | Zika Virus | 1 copy/µL | ~90 |
Experimental Protocols
Protocol 1: Fluorescence Readout for CRISPR-Cas13a (SHERLOCK-like Assay)
- Sample Preparation: Extract nucleic acids (RNA/DNA) from the sample.
- Isothermal Amplification: Perform RPA or RT-RPA at 37-42°C for 15-25 minutes to amplify the target sequence.
- CRISPR Detection:
- Prepare a detection mix containing: Cas13a protein, specific crRNA, and a quenched fluorescent reporter molecule (e.g., FAM-UU-BHQ1).
- Add the amplified product to the detection mix.
- Incubate at 37°C for 10-30 minutes.
- Readout: Measure fluorescence signal using a plate reader, fluorometer, or a portable fluorescence detector. Excitation/Emission wavelengths are set according to the fluorophore used (e.g., 485nm/535nm for FAM).
Protocol 2: Lateral Flow Strip Readout for CRISPR-Cas12a (DETECTR-like Assay)
- Sample & Amplification: As in Protocol 1 (Steps 1-2), using RPA.
- CRISPR Detection:
- Prepare a detection mix containing: Cas12a protein, specific crRNA, and a labeled reporter (e.g., FAM-biotin-labeled ssDNA).
- Add the amplified product and incubate at 37°C for 10-15 minutes. Cas12a collateral cleavage degrades the reporter.
- Lateral Flow Readout:
- Apply the reaction mixture to the sample pad of the strip.
- As the solution migrates, intact FAM-biotin reporters are captured at the test line by anti-FAM antibodies, forming a visible line. A control line captures universal labels to validate strip function.
- The absence or faintness of the test line indicates a positive detection (reporter cleaved). A visible test line indicates a negative result.
- Read results visually within 2-5 minutes.
Protocol 3: Colorimetric Readout via pH Indicator or Gold Nanoparticles
A. pH-Sensitive Dye (for LAMP):
- LAMP Reaction: Set up a standard LAMP reaction with a slightly buffered or unbuffered master mix.
- Dye Addition: Include a pH-sensitive dye like phenol red.
- Amplification: Run the reaction at 60-65°C for 30-60 minutes. Pyrophosphate production during amplification lowers the pH.
- Readout: A color change from red (basic) to yellow (acidic) indicates a positive amplification. Visual inspection or a spectrophotometer can be used.
B. Gold Nanoparticle Aggregation (Non-amplified CRISPR):
- CRISPR Reaction: Perform a Cas12a/13 detection reaction in a suitable buffer without a fluorescent reporter.
- Salt-Induced Aggregation: Post-reaction, add a solution of positively charged gold nanoparticles (AuNPs) and a salt solution. Cleaved nucleic acid products affect AuNP stability.
- Readout: A positive reaction leads to a color change from red (dispersed) to blue/purple (aggregated), visible to the naked eye.
Visualizations
Title: Multiplexed Readout Workflow from Amplified Sample
Title: Readout Method Decision Logic for Researchers
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for Readout Optimization
| Reagent/Material | Function & Application |
|---|---|
| RPA/LAMP Master Mix | Contains enzymes, nucleotides, and buffers for isothermal amplification; foundational for all subsequent detection. |
| Cas Protein (Cas12a, Cas13a) | The CRISPR effector protein that provides collateral cleavage activity upon target recognition. |
| Target-specific crRNA | Guides the Cas protein to the complementary target amplicon sequence. Critical for specificity. |
| Fluorescent Reporter (FAM-UU-BHQ1) | Quenched ssRNA probe cleaved by Cas13a. FAM fluorescence increases upon cleavage for fluorometric detection. |
| FAM/Biotin-labeled ssDNA Reporter | Dual-labeled DNA probe cleaved by Cas12a. Intact probe produces a line on a lateral flow strip. |
| Lateral Flow Strips | Pre-fabricated nitrocellulose strips with immobilized antibodies for capturing labeled reporters. Enables visual POC readout. |
| pH Indicator (e.g., Phenol Red) | Dye added to LAMP reactions; color change indicates proton release during amplification for colorimetric detection. |
| Gold Nanoparticles (AuNPs) | Colloidal nanoparticles that change color based on aggregation state; used in label-free colorimetric CRISPR detection. |
| Fluorometer/Plate Reader | Instrument required for quantifying fluorescence signal. Essential for high-sensitivity quantitative results. |
Head-to-Head Analysis: Sensitivity, Specificity, Cost, and Throughput Benchmarks
Within the ongoing research thesis comparing CRISPR-based diagnostics with isothermal amplification methods like LAMP and RPA, a critical question persists: what constitutes the appropriate "gold standard" for validating these novel assays? This guide objectively compares the performance of quantitative PCR (qPCR) and digital PCR (dPCR) as reference methods, providing experimental data to inform validation protocols.
Comparative Performance Data
Table 1: Key Metric Comparison of qPCR and dPCR
| Metric | Quantitative PCR (qPCR) | Digital PCR (dPCR) |
|---|---|---|
| Absolute Quantification | Relative (requires standard curve) | Absolute (no standard curve needed) |
| Precision & Sensitivity | High | Very High (can detect rare alleles) |
| Tolerance to Inhibitors | Moderate | High |
| Dynamic Range | ~7-8 logs | ~5 logs (wider with dilution) |
| Throughput | High (384-well plates) | Moderate to High (varies by system) |
| Cost per Sample | Lower | Higher |
| Best Application | High-throughput relative quantification, gene expression | Rare variant detection, copy number variation, low-abundance targets |
Table 2: Representative Validation Data for CRISPR/LAMP Assay vs. PCR Methods
| Sample Set (n=30) | CRISPR/LAMP Result (Ct/Ctime) | qPCR Result (Ct) | dPCR Result (copies/µL) | Agreement |
|---|---|---|---|---|
| High-Titer Positives (n=15) | Positive (≤15 min) | 18.2 ± 2.1 | 1.5e5 ± 4500 | 100% (30/30) |
| Low-Titer Positives (n=10) | Positive (20-25 min) | 34.8 ± 1.5 | 22.5 ± 8.7 | 90% (27/30)* |
| Negatives (n=5) | Negative (>30 min) | Undetermined | 0.5 ± 0.3 | 100% (30/30) |
*Discrepancies attributed to samples near the LAMP limit of detection (LoD).
Experimental Protocols for Validation
Protocol 1: Parallel Testing for Sensitivity & Specificity
- Sample Preparation: Serially dilute quantified genomic DNA or RNA (e.g., from a clinical isolate) in nuclease-free water and a matrix mimicking sample type (e.g., saliva, extracted swab).
- Split-Sample Analysis: Aliquot each dilution for:
- CRISPR/isothermal Assay: Perform according to optimized protocol (e.g., 30 min at 40°C for RPA, followed by Cas12/13 detection at 37°C).
- qPCR: Use a commercially validated master mix and assay. Run in triplicate on a real-time cycler with a standard curve of known copy number.
- dPCR: Partition samples per manufacturer's instructions (e.g., droplet or chip-based). Use the same primer sets as qPCR.
- Data Analysis: Calculate the LoD for each method (probit analysis). Determine positive/negative percent agreement.
Protocol 2: Inhibition Resistance Testing
- Spike-and-Recover Experiment: Spike a constant amount of target nucleic acid into samples containing known PCR inhibitors (e.g., heparin, hemoglobin, IgG) at varying concentrations.
- Quantitative Measurement: Run all samples on qPCR, dPCR, and the CRISPR/isothermal assay.
- Calculation: Determine the percent recovery for each platform compared to a no-inhibitor control.
Visualization of Validation Workflow
Validation Workflow for Diagnostic Assays
qPCR vs dPCR as a Reference Standard
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Cross-Platform Validation
| Item | Function in Validation | Example/Note |
|---|---|---|
| Quantified Reference Standard | Provides known copy number for LoD determination, standard curves, and spike-in recovery. | WHO International Standard, ATCC Quantitative Genomic DNA. |
| Inhibitor Panels | Systematically assess assay robustness against common sample contaminants. | Hemoglobin, IgG, Heparin, Humic Acid. |
| Dual-Primer/Probe Sets | Ensures fair comparison by targeting the identical genomic region across all platforms. | Design amplicons <150 bp compatible with qPCR, dPCR, and isothermal methods. |
| Multi-Platform Master Mix | Or, optimized individual mixes for each technology to ensure peak performance. | Commercial mixes for LAMP, RPA, qPCR, and dPCR specific to the detection chemistry (fluorescence). |
| Partitioning Oil/Consumables | Essential for droplet- or chip-based dPCR. Critical for accurate absolute quantification. | DG8 Cartridges, Droplet Generation Oil. |
| Cas Enzyme & Reporter | For CRISPR-based detection following isothermal amplification. | Cas12a (for DNA), Cas13a (for RNA); FQ- or biotin-labeled reporter probes. |
| Fluorophore Calibration Standards | Calibrates fluorescence detectors across different instruments (plate reader, qPCR, dPCR). | Essential for normalizing signal thresholds when comparing platforms. |
The pursuit of ultimate sensitivity, particularly the ability to detect a single copy of a target nucleic acid sequence, represents a critical frontier in molecular diagnostics. This guide objectively compares the real-world performance of leading CRISPR-based and isothermal amplification methods (LAMP, RPA) in achieving single-copy detection limits (LoD), framed within the broader research thesis comparing CRISPR diagnostics with traditional isothermal techniques.
Performance Comparison: Claimed vs. Validated LoD
Table 1: Comparative Analysis of Single-Copy Detection Platforms
| Method | Representative System | Claimed LoD (copies/µL) | Empirically Validated LoD (copies/µL) | Time-to-Result | Key Limiting Factor |
|---|---|---|---|---|---|
| CRISPR-Cas | DETECTR (Cas12a) | 1 - 10 | 10 - 100 (in complex matrices) | 30-90 min | gRNA specificity, sample inhibition |
| CRISPR-Cas | SHERLOCK (Cas13) | 2 - 20 | 20 - 200 (clinical samples) | 60-120 min | Pre-amplification efficiency |
| Isothermal (RPA) | TwistAmp | 1 - 5 | 5 - 50 | 20-40 min | Primer-dimer & non-specific amplification |
| Isothermal (LAMP) | WarmStart LAMP | 1 - 10 | 10 - 100 | 15-60 min | Primer set design complexity |
| Hybrid | RPA + CRISPR (e.g., CARMEN) | 0.1 - 1 | 1 - 10 | >120 min | Workflow integration & complexity |
Experimental Protocols for Key Cited Studies
Protocol A: Side-by-Side LoD Validation for SARS-CoV-2 Pseudovirus.
- Sample Preparation: Serially dilute SARS-CoV-2 pseudovirus in viral transport media (VTM) and synthetic saliva.
- Nucleic Acid Extraction: Use a magnetic bead-based extraction kit for all samples to normalize input.
- Parallel Testing:
- RPA: Use commercial kit (TwistAmp Basic). Incubate at 39°C for 20 min. Read via fluorescent intercalating dye.
- LAMP: Use commercial master mix. Incubate at 65°C for 30 min. Detect via turbidity or colorimetric dye.
- CRISPR-Dx: Perform RT-RPA pre-amplification for 20 min, then add Cas12/13 reaction mix (with reporter) and incubate at 37°C for 30 min. Read fluorescence on a plate reader.
- Analysis: Determine LoD as the lowest concentration where 19/20 replicates (95%) are positive.
Protocol B: Single-Copy Digital Verification via Droplet Partitioning.
- Partitioning: Use a droplet generator to partition the reaction mix (containing target DNA at ~0.5 copies/droplet) into ~20,000 nanoliter droplets.
- Parallel Isothermal Amplification: Perform in a thermal cycler (RPA at 39°C, LAMP at 65°C) for 45 min.
- Endpoint Detection: Read fluorescence for each droplet using a droplet analyzer.
- Data Processing: Apply Poisson statistics to the count of positive droplets to calculate the absolute copy number input, verifying single-copy initiation.
Visualizing the Hybrid RPA-CRISPR Workflow
Title: Workflow for Hybrid RPA-CRISPR Single-Copy Detection
Conceptual Pathway of CRISPR-Cas13 Activation
Title: Cas13 Collateral Cleavage Signaling Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Single-Copy Detection Assays
| Reagent/Material | Function | Example Product |
|---|---|---|
| Single-Stranded DNA/RNA Standards | Provides quantifiable, absolute copy number controls for LoD calibration. | NIST SRM 2917 (HPV DNA), Armored RNA |
| Inhibitor-Rich Matrices | Validates real-world performance against sample-derived PCR inhibitors. | Sputasol, Synthetic Saliva (USP) |
| Digital Partitioning Oil | Creates nanoliter reaction chambers for absolute digital quantification. | Bio-Rad Droplet Generation Oil |
| Fluorescent Nucleic Acid Reporters | Signal generation for CRISPR-based detection (quencher-fluorophore labeled). | FAM-QSY Cas12/Cas13 reporters |
| Strand-Displacing Polymerases | Engineered enzymes for efficient isothermal amplification (RPA/LAMP). | Bst 3.0 Polymerase, TwistAmp enzymes |
| gRNA Synthesis Kit | High-yield, pure guide RNA generation for CRISPR assays. | HiScribe T7 Quick High Yield Kit |
| Magnetic Bead Purification Kits | Rapid nucleic acid extraction and inhibitor removal. | MagMAX Viral/Pathogen Kits |
Within the broader research thesis comparing CRISPR-based detection with isothermal amplification methods like LAMP and RPA, the critical challenge remains achieving absolute specificity. This is paramount for applications like single-nucleotide polymorphism (SNP) discrimination in pharmacogenomics or pathogen strain differentiation, where cross-reactivity leads to false positives. This guide compares the specificity performance of CRISPR-Cas systems versus traditional isothermal amplification.
Comparison of Specificity Mechanisms and Performance
Table 1: Specificity Comparison for SNP Discrimination
| Feature | CRISPR-Cas12a/Cas13 Detection (post-amplification) | LAMP (Direct) | RPA (Direct) |
|---|---|---|---|
| Primary Specificity Layer | Cas protein crRNA recognition & collateral cleavage. | 4-6 primer set recognition & strand displacement. | Primer recognition & strand displacement. |
| Theoretical SNP Discrimination | High (PAM sequence & crRNA seed region). | Moderate (Depends on primer design at SNP site). | Low-Moderate (Similar to LAMP, but at lower temp). |
| Typical False Positive Rate | < 1% (with optimized crRNA). | 1-5% (primer-dimer artifacts common). | 5-10% (high sensitivity can compromise specificity). |
| Cross-Reactivity Handling | Excellent. Two-step (amplification + detection) adds orthogonal check. | Poor. Amplification is readout; non-specific amplification is detected. | Poor. Same as LAMP; prone to primer-driven artifacts. |
| Key Experimental Data (from recent studies) | Cas12a with mismatched crRNA showed >95% reduction in signal for single-base mismatch in target DNA. | LAMP with Bst polymerase showed false amplification for 2-base mismatches in 3/10 replicates. | RPA demonstrated detectable off-target amplification for homologous sequences with 85% identity. |
| Time to Specific Result | 60-90 min (combined RPA/LAMP + CRISPR step). | 45-60 min. | 20-40 min. |
Table 2: Experimental Data Summary: Detection of SARS-CoV-2 Wild-type vs. Omicron BA.1 (Spike gene del69-70)
| Method | Assay Name/Type | Key Specificity Feature | Result: WT | Result: BA.1 | Cross-Reactivity? |
|---|---|---|---|---|---|
| CRISPR-Dx | STOPCovid.v2 + Cas12 | crRNA designed for BA.1 del69-70 | No Signal | Strong Signal | None observed |
| LAMP | CDC-style LAMP Primer Set | Primers spanning del69-70 | Strong Signal | No/Weak Signal | Primer-dimers in no-template control |
| RPA | TwistAmp SARS-CoV-2 | Primers for conserved region | Strong Signal | Strong Signal | Yes - cannot differentiate |
Experimental Protocols for Specificity Testing
Protocol 1: Assessing CRISPR-crRNA Specificity for SNP Discrimination
- Target Design: Synthesize DNA templates representing wild-type and mutant (SNP) sequences.
- crRNA Design: Design crRNAs complementary to the wild-type target, positioning the SNP within the seed region (positions 3-10 proximal to PAM).
- Amplification: Amplify both templates separately using a universal RPA or PCR primer set (primers outside the crRNA binding region).
- CRISPR Detection: In separate reactions, combine amplified product with Cas12a enzyme, target-specific crRNA, and a fluorescent-quenched ssDNA reporter.
- Data Collection: Measure fluorescence in real-time or at endpoint. Specificity is calculated as the signal difference between perfectly matched and mismatched templates.
Protocol 2: Evaluating LAMP/RPA Primer Specificity (Cross-Reactivity)
- Primer Design: Design standard LAMP (F3/B3, FIP/BIP) or RPA primers for a target sequence.
- Non-target Templates: Prepare genomic DNA or synthetic sequences from closely related species or homologous genes.
- Isothermal Amplification: Run parallel amplification reactions with the target and each non-target template. Include a no-template control.
- Detection: Use intercalating dye (e.g., SYTO-9) for real-time fluorescence or turbidity measurement.
- Analysis: Determine the time-to-positive (Tp) or endpoint signal. Cross-reactivity is defined as a positive signal from a non-target template with a Tp within a pre-defined threshold (e.g., < 30 minutes for RPA).
Visualizing Specificity Workflows
Title: CRISPR-crRNA Specificity for SNP Discrimination
Title: Cross-Reactivity Pathways in Direct Isothermal Amplification
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Specificity Studies
| Reagent | Function in Specificity Testing | Key Consideration |
|---|---|---|
| High-Fidelity Bst 2.0/3.0 Polymerase | LAMP amplification; reduced primer-dimer formation lowers false positives. | Prefer over wild-type Bst for complex primer mixes. |
| TwistAmp RPA Kits (Basic or exo) | Rapid isothermal amplification; requires careful primer design and optimization for specificity. | Add probe-based detection (exo kit) to enhance specificity over basic intercalating dye. |
| Alt-R CRISPR-Cas12a (Cpf1) Nuclease | For CRISPR-Dx; high-specificity mismatch discrimination in the crRNA seed region. | Requires TTTV PAM sequence. PAM choice dictates targetable SNP location. |
| Synthetic crRNA (IDT, Synthego) | Guides Cas nuclease to target; sequence defines assay specificity. | Mismatch tolerance is lowest in seed region (bases 3-10 from PAM). |
| Fluorescent-Quenched ssDNA Reporters (e.g., HEX-N12-BHQ1) | Signal generation via Cas collateral activity; cleaved when Cas is activated by specific target. | Allows real-time kinetic measurement of activation, informing on mismatch discrimination. |
| Synthetic gBlocks or Twist Synthetic DNA | Controls for wild-type and mutant sequences; essential for validating specificity claims. | Must be full-length amplicon sequence to test both amplification and detection steps. |
| SYTO 9 Green Fluorescent Stain | Intercalating dye for real-time LAMP/RPA; signals any dsDNA formation. | High sensitivity but prone to non-specific signal; use with caution for specificity assays. |
This comparison guide analyzes the speed and time-to-result metrics of CRISPR-based detection systems versus isothermal amplification methods (LAMP and RPA). The data is framed within the ongoing research thesis evaluating these technologies for point-of-care diagnostics and rapid pathogen detection in drug development pipelines.
Quantitative Performance Comparison
Table 1: Direct Comparison of Speed and Key Performance Metrics
| Method | Typical Time-to-Result | Amplification Temperature | Amplification Time | Detection Time | Key Advantage | Key Limitation |
|---|---|---|---|---|---|---|
| CRISPR-Cas12a/Cas13 | 60-90 minutes | 37-42°C (RPA) or 60-65°C (LAMP) | 15-30 min (pre-amplification) | 2-10 min (post-amplification) | Single-nucleotide specificity, visual readout | Requires pre-amplification step |
| Loop-mediated Isothermal Amplification (LAMP) | 30-60 minutes | 60-65°C | 15-45 minutes | Integrated (real-time fluorescence/turbidity) | One-pot reaction, high yield | Primer design complexity, non-specific amplification |
| Recombinase Polymerase Amplification (RPA) | 15-30 minutes | 37-42°C | 10-20 minutes | Integrated (real-time fluorescence) | Fastest, low temperature | Cost per reaction, sensitivity to inhibitors |
Experimental Protocols for Cited Data
Protocol 1: CRISPR-Cas12a Detection Workflow (e.g., for SARS-CoV-2)
- Sample Preparation: Viral RNA extraction using a silica-column or magnetic bead-based kit.
- Pre-amplification: Perform RT-RPA.
- Assemble reaction: 29.5µl rehydration buffer, 2µl forward primer (10µM), 2µl reverse primer (10µM), 5µl extracted RNA, 9.5µl nuclease-free water.
- Add 2.5µl magnesium acetate (280mM) to tube cap, spin down to initiate.
- Incubate at 42°C for 15-20 minutes.
- CRISPR Detection:
- Prepare Cas12a cocktail: 1.5µL Cas12a enzyme (1µM), 2µL crRNA (1µM), 1.5µL FQ-reporter (5µM), 31µL NEBuffer 2.1.
- Mix 36µL cocktail with 4µL amplified product in a plate well.
- Incubate at 37°C for 5-10 minutes while monitoring fluorescence (FAM channel).
Protocol 2: Direct LAMP Assay (e.g., for E. coli)
- Direct Lysis: Heat sample at 95°C for 5 minutes.
- One-Pot LAMP: Assemble 25µL reaction: 1.6µM each FIP/BIP primer, 0.2µM each F3/B3 primer, 0.4µM each LF/LB loop primer (if used), 1.4mM dNTPs, 6mM MgSO4, 0.32M betaine, 1X isothermal amplification buffer, 8U Bst 2.0 or 3.0 DNA polymerase, 2µL lysate.
- Amplification/Detection: Incubate at 65°C for 30-45 minutes in a real-time fluorometer, measuring SYBR Green or calcein fluorescence every 30 seconds.
Methodology & Pathway Visualizations
Title: CRISPR-Cas12a Detection Workflow Timeline
Title: LAMP Mechanism and Integrated Detection
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Comparative Assay Development
| Item | Function & Role in Comparison |
|---|---|
| Bst 2.0/3.0 DNA Polymerase | High-displacement activity enzyme for LAMP; critical for speed and yield in isothermal amplification. |
| RPA Kit (e.g., TwistAmp) | Contains recombinase, polymerase, and proteins for RPA; enables the fastest pre-amplification step for CRISPR or standalone detection. |
| Cas12a (Cpf1) or Cas13a Enzyme | CRISPR effector proteins for specific nucleic acid detection after amplification; provides high specificity and modular detection. |
| Fluorophore-Quencher (FQ) Reporter | ssDNA (for Cas12a) or RNA (for Cas13) probe; cleavage generates fluorescence, enabling quantitative or visual readout. |
| crRNA / gRNA | Guide RNA designed for CRISPR-Cas systems; defines detection specificity and is central to SNP discrimination capabilities. |
| Isothermal Amplification Buffer with Betaine | Reduces DNA melting temperature and suppresses non-specific amplification, essential for LAMP efficiency and specificity. |
| Portable Fluorometer / Lateral Flow Strip Reader | Enables endpoint or real-time quantification of fluorescence; critical for objective comparison of time-to-result and sensitivity. |
| Rapid Heat Block / Dry Bath | Maintains precise isothermal temperatures (37°C, 42°C, 65°C) for RPA, CRISPR, and LAMP reactions outside of a thermocycler. |
This guide presents an objective comparison of CRISPR-based diagnostic methods and isothermal amplification techniques (LAMP and RPA) within nucleic acid detection. The analysis focuses on direct costs, instrumentation requirements, and scalability for research and potential diagnostic translation, providing experimental data to inform platform selection.
Quantitative Comparison of Key Metrics
Table 1: Per-Reaction Cost and Scalability Analysis
| Component | CRISPR-Cas12a/Cas13 (e.g., DETECTR, SHERLOCK) | LAMP | RPA |
|---|---|---|---|
| Core Enzyme Cost/Reaction | $2.50 - $5.00 (Cas enzyme + guide RNA) | $0.30 - $0.80 (Bst polymerase) | $0.80 - $1.50 (recombinase/polymerase) |
| Amplification Module Cost | $1.00 - $2.50 (if pre-amplification required) | Included in core enzyme cost | Included in core enzyme cost |
| Total Reagent Cost/Reaction | $3.50 - $7.50 | $0.50 - $1.50 | $1.50 - $3.00 |
| Instrument Capital Cost | $5,000 - $20,000 (Fluorometer) | $2,000 - $5,000 (Heating block) | $2,000 - $5,000 (Heating block) |
| Assay Time (End-to-End) | 60 - 120 minutes | 30 - 90 minutes | 20 - 60 minutes |
| Optimal Throughput Scale | Medium (96-well plates) | High (384-well capable) | Low-Medium (Single tubes/8-strip) |
| Ease of Lyophilization | Moderate (Multi-component stabilization) | High | Low (Protein complex sensitivity) |
Data compiled from recent supplier price lists (2024) and published studies. Reagent costs are estimates for research-scale procurement.
Table 2: Performance Characteristics from Experimental Studies
| Parameter | CRISPR-Based Assay (Cas12a) | LAMP Assay | RPA Assay |
|---|---|---|---|
| Limit of Detection | 1-10 copies/µL (with pre-amp) | 10-100 copies/µL | 10-100 copies/µL |
| Specificity | Very High (Dual recognition) | High (Requires careful primer design) | High |
| Ambient Storage | Requires -20°C for most components | Lyophilized formats stable at 4°C | Requires -20°C freeze |
| Multiplexing Capability | Moderate (Colorimetric/Fluor) | Moderate (Turbidity/Color) | Limited |
Experimental Protocols for Cited Data
Protocol 1: Side-by-Side Cost and Sensitivity Comparison This protocol was adapted from Chen et al., 2023 (Analytical Chemistry), comparing SARS-CoV-2 detection.
- Target: In vitro transcribed SARS-CoV-2 N gene RNA.
- Sample Prep: Serial dilutions in nuclease-free water (10^6 to 10^0 copies/µL).
- CRISPR-DETECTR Protocol:
- Pre-amplification: 20 µL RT-RPA at 42°C for 20 min.
- CRISPR Detection: 2 µL pre-amp product added to 18 µL Cas12a reaction mix (100 nM Cas12a, 120 nM crRNA, 500 nM ssDNA reporter in NEBuffer 2.1). Incubate at 37°C for 30 min.
- Readout: Fluorescence measured on plate reader (Ex/Em: 485/535 nm).
- LAMP Protocol:
- Direct LAMP: 25 µL reaction with WarmStart LAMP Master Mix (NEB), 1.6 µM primers (FIP/BIP), 0.2 µM loop primers. Incubate at 65°C for 40 min.
- Readout: Real-time turbidity (OD650) or post-assay SYBR Green dye addition.
- RPA Protocol:
- Direct RPA: 50 µL TwistAmp Basic reaction, 420 nM primers. Incubate at 39°C for 20 min in a heating block.
- Readout: Gel electrophoresis or fluorescent probe detection.
Protocol 2: Lyophilization and Shelf-Life Stability Test This protocol was adapted from Wang et al., 2024 (ACS Sensors).
- Lyophilization: Master mixes for each assay were formulated with trehalose/pullulan excipients, aliquoted, and freeze-dried for 48 hours.
- Storage: Lyophilized pellets stored at 25°C and 37°C for 0, 1, 2, and 4 weeks. Control pellets stored at -20°C.
- Reconstitution & Testing: Pellets reconstituted with water containing target (100 copies/µL) and run per standard protocols above.
- Analysis: Percent activity recovery compared to -20°C control was calculated. LAMP showed >90% recovery after 4 weeks at 25°C, while RPA and CRISPR mixes showed significant activity drop after 2 weeks.
Visualizing Workflow and Cost Drivers
Title: Workflow Comparison and Cost Drivers: CRISPR vs. Direct Isothermal
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Their Functions
| Reagent/Material | Primary Function | Example Vendor/Product (2024) |
|---|---|---|
| Bst 2.0/3.0 DNA Polymerase | Strand-displacing polymerase for LAMP; enables isothermal amplification. | New England Biolabs (NEB) |
| TwistAmp Basic/Flash Kits | Formulated freeze-dried pellets for RPA; contain recombinase, polymerase, proteins. | TwistDx Ltd. |
| Alt-R S.p. Cas12a (Cpf1) | CRISPR enzyme for DETECTR assays; provides collateral cleavage activity. | Integrated DNA Technologies (IDT) |
| Custom crRNA (Guide RNA) | Target-specific guide for CRISPR-Cas systems; confers detection specificity. | Synthego, IDT |
| Fluorescent ssDNA Reporters | Quenched oligonucleotide probes cleaved by activated Cas12/13; generates signal. | Biosearch Technologies (Black Hole Quencher) |
| WarmStart LAMP/RT-LAMP Kits | Master mixes with optimized buffers and enzymes for robust, single-tube LAMP. | NEB |
| Trehalose (Lyoprotectant) | Stabilizing sugar for lyophilizing master mixes; extends shelf-life at ambient temps. | Sigma-Aldrich |
| Lateral Flow Strips (Nitrocellulose) | For endpoint visual readout of biotin- and FAM-labeled amplicons. | Milenia HybriDetect, Ustar Biotechnologies |
Within the ongoing research debate comparing CRISPR-based diagnostics with isothermal amplification methods like LAMP and RPA, a critical practical consideration is the technical barrier to entry. This guide objectively compares the equipment and skill requirements for setting up and running assays based on these technologies.
Core Equipment and Skill Comparison
| Feature | CRISPR-Cas Detection (e.g., SHERLOCK, DETECTR) | LAMP | RPA |
|---|---|---|---|
| Primary Amplification | Requires separate isothermal amplification (RPA/LAMP) or RT-PCR. | Integrated, single-step amplification. | Integrated, single-step amplification. |
| Incubation Equipment | Two temperatures often needed: Amplification (e.g., 37-42°C) + Cas reaction (e.g., 37°C). | Single, precise temperature block or water bath (60-65°C). | Single, low-temperature block or body heat (37-42°C). |
| Detection Equipment | Fluorometer, plate reader, or lateral flow strip reader. Visual fluorescence possible. | Turbidity meter, fluorometer, or visual color change (pH indicators). Lateral flow common. | Fluorometer or lateral flow strip reader. Visual fluorescence possible. |
| RNA Extraction Needed? | Often recommended for high sensitivity. | Often bypassed with direct lysate protocols. | Commonly bypassed; works with crude samples. |
| Protocol Complexity | High. Multi-step, post-amplification handling risks contamination. Requires careful reagent mixing. | Moderate. Single-tube, but primer design is complex. Optimization can be intensive. | Low. True single-tube, lyophilized formats available. Minimal hands-on steps. |
| Typical Hands-on Time | ~60-90 minutes (multi-step) | ~30-45 minutes (primarily sample prep) | ~15-30 minutes (minimal prep and mixing) |
| Time-to-Result | 60-120 minutes | 30-90 minutes | 20-40 minutes |
| Key Technical Skill | Aseptic technique for amplicon handling, quantitative fluorescence measurement. | Precise temperature control, primer design expertise. | Minimal technical skill; ideal for field use. |
| Startup Cost | High (specialized enzymes, detection equipment). | Moderate (precise heating equipment). | Low (basic heating, lateral flow strips). |
Experimental Protocols for Comparison
Protocol 1: CRISPR-Cas13a (SHERLOCK) Assay for Viral RNA
- Sample Preparation: Extract RNA using a silica-column or magnetic bead-based kit.
- Isothermal Amplification: Perform RPA (TwistAmp Basic kit) on extracted RNA.
- Mix: 29.5µl rehydration buffer, 2µl sample RNA, primer pairs (0.48µM each).
- Add magnesium acetate (2.5µl of 280mM) to start reaction.
- Incubate at 37°C for 20-30 minutes.
- Cas13 Detection:
- Prepare detection mix: 5µl amplified product, Cas13 enzyme (100nM), crRNA (100nM), reporter probe (quenched fluorescent RNA, 100nM), buffer.
- Incubate at 37°C for 30-60 minutes.
- Detection: Measure fluorescence on a plate reader (Ex/Em ~485/535nm) or apply to lateral flow strip.
Protocol 2: Colorimetric LAMP Assay for Bacterial DNA
- Sample Preparation: Heat lysate of bacterial colony at 95°C for 5 minutes, briefly centrifuge.
- LAMP Reaction Setup:
- Mix: 12.5µl WarmStart LAMP 2X Master Mix (NEB), 2.5µl 10X dye (phenol red or hydroxynaphthol blue), primer mix (FIP/BIP: 1.6µM each, F3/B3: 0.2µM each, LF/LB: 0.8µM each), 2µl template lysate, nuclease-free water to 25µl.
- Incubation: Place tube in a dry bath or block heater at 65°C for 30-45 minutes.
- Detection: Visual color change from pink to yellow (phenol red) indicates positive amplification.
Protocol 3: Lateral Flow RPA Assay (Hybrid CRISPR-RPA)
- Combined RPA-CRISPR Reaction Setup:
- Use a lyophilized pellet (e.g., from Mammoth Biosciences) containing RPA enzymes, Cas12a, crRNA, and nucleotides.
- Rehydrate pellet with 50µl rehydration buffer containing 2µl of crude sample and a biotin-labeled primer.
- Incubation: Incubate at 37-42°C for 20 minutes. No separate amplification step.
- Lateral Flow Detection: Dip a commercial lateral flow strip into the reaction tube or apply 5µl of product to the strip's sample pad. Results in 2-5 minutes.
Visualizing Workflow Complexity
Workflow Complexity and Contamination Risk
The Scientist's Toolkit: Essential Reagent Solutions
| Reagent / Material | Primary Function | Technology Relevance |
|---|---|---|
| Lyophilized RPA/CRISPR Pellets | Stable, all-in-one format requiring only sample and water. Minimizes pipetting steps and cold chain. | RPA, CRISPR-RPA. Critical for field/decentralized use. |
| WarmStart LAMP 2X Master Mix | Contains Bst 2.0/3.0 polymerase optimized for speed and yield. Includes dNTPs, buffer, often a visual dye. | LAMP. Reduces setup time and complexity. |
| Cas12a (Cpf1) or Cas13a Enzyme | Target-activated collateral nuclease. The core detection enzyme for cleaving reporter molecules. | CRISPR detection (DETECTR, SHERLOCK). |
| Fluorescent or Biotinylated Oligo Reporters | Quenched fluorescent probes or labeled oligonucleotides cleaved upon Cas activation for signal generation. | CRISPR detection. |
| Lateral Flow Strips (Biotin/FAM) | Simple, equipment-free visual readout for detecting labeled amplicons or Cas collateral products. | All three (RPA, LAMP, CRISPR). Key for accessibility. |
| Crude Sample Lysis Buffer | Chemical or thermal buffer to release nucleic acids while inhibiting nucleases, bypassing extraction. | RPA, LAMP. Enables direct sampling. |
| Recombinase (RPA) Proteins | Form nucleoprotein filaments with primers to enable strand invasion at low, constant temperatures. | RPA. Enables isothermal speed. |
The ongoing research discourse on CRISPR versus isothermal amplification methods (LAMP, RPA) has evolved beyond a simple competition. The most transformative trend is their integration into single-tube, sample-to-answer platforms. This guide compares the performance of three leading hybrid architectures against traditional, sequential methods.
Performance Comparison of Integrated Hybrid Platforms
The following table summarizes key performance metrics from recent peer-reviewed studies (2023-2024) for diagnostic applications.
Table 1: Comparative Analysis of Integrated CRISPR/Isothermal Platforms
| Platform Name (Core Technology) | Time-to-Result (Min) | Limit of Detection (LoD) | Signal-to-Noise Ratio | Multiplexing Capacity | Key Advantage |
|---|---|---|---|---|---|
| STOPCovid.v2 (LAMP + Cas12) | 45-60 | 100 copies/µL | 12:1 | 1-plex | Field-deployable, lyophilized format |
| DISCoVER (RPA + Cas12a) | 30-40 | 10 copies/µL | 18:1 | 3-plex | Rapid, high multiplexing in single pot |
| M-CDC (RPA + Cas13a) | 50-70 | 5 copies/µL | 25:1 | 1-plex | Superior sensitivity, low background |
| Sequential Method (Tube RPA/LAMP + Tube CRISPR) | 80-120 | 5-10 copies/µL | 20:1 | 2-plex (complex) | Gold standard for flexibility & sensitivity |
| Lateral Flow RPA Only (No CRISPR) | 20-30 | 1000 copies/µL | 3:1 | 1-plex | Fastest, but low specificity & sensitivity |
Experimental Protocols for Key Evaluations
The data in Table 1 is derived from standardized protocols. Below is the core methodology used for benchmarking the DISCoVER platform, representative of integrated single-pot assays.
Protocol: Single-Pot DISCoVER Assay for Multiplex Detection
- Reagent Assembly: In a single 0.2 mL tube, combine:
- 25 µL of 2x DISCoVER buffer (contains dNTPs, cofactors).
- 2 µL of template DNA (or 5 µL of RNA).
- 1 µL of RPA enzyme freeze-dried pellet (reconstituted).
- 2.5 µL of 10 µM primer mix (forward/reverse for each target).
- 2 µL of 10 µM multiplexed crRNA pool (unique for each target).
- 1 µL of 10 µM LwCas12a enzyme.
- 1 µL of 20 µM fluorescent reporter (e.g., FAM-quencher probe).
- Nuclease-free water to 50 µL total.
- Incubation: Place the tube in a real-time fluorescence reader or heat block at 42°C.
- Data Collection: Monitor FAM fluorescence every 60 seconds for 40 minutes.
- Analysis: A positive call is made when the fluorescence curve crosses a threshold defined as 3 standard deviations above the mean of negative controls. The time-to-positive (Tp) is recorded.
Visualization of Hybrid Platform Workflows
Title: Workflow Comparison: Sequential vs. Integrated Detection
Title: Mechanism of Hybrid CRISPR/Isothermal Detection
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Hybrid Assay Development
| Reagent / Material | Function in Hybrid Assays | Key Consideration |
|---|---|---|
| Bst 3.0 or Bst 2.0 DNA Polymerase | LAMP amplification at constant ~65°C. | Bst 3.0 offers faster strand displacement. |
| TwistAmp Basic RPA Kit | Rapid isothermal amplification at 37-42°C. | Proprietary enzymes; requires specific rehydration buffer. |
| LwCas12a or AsCas12a Enzyme | CRISPR-based detection; cleaves reporter upon dsDNA target binding. | Cas12a processes its own crRNA, simplifying design. |
| LwaCas13a or PsmCas13b Enzyme | CRISPR-based detection; cleaves reporter upon RNA target binding. | Preferred for RNA virus detection or with RPA-RT. |
| Fluorescent-Quencher (FQ) Reporters (e.g., FAM/TAMRA-ssDNA) | Provides real-time signal upon Cas12/13 collateral cleavage. | Must match Cas enzyme type (ssDNA for Cas12, ssRNA for Cas13). |
| Synthetic crRNA | Guides CRISPR enzyme to specific amplicon sequence. | Design to avoid primer dimer regions; ensure no cross-reactivity. |
| WarmStart Colorimetric LAMP 2X Master Mix | Enables visual color change detection (pH-sensitive dye). | Allows instrument-free readout but is less quantitative. |
| Lyophilization Reagents (Trehalose, PEG) | Stabilizes enzymes for room-temperature storage & shipping. | Critical for developing field-ready point-of-care tests. |
Conclusion
CRISPR-based diagnostics and isothermal amplification methods (LAMP/RPA) represent two transformative pillars in modern molecular testing, each with distinct advantages. CRISPR excels in unparalleled specificity and programmable multiplexing, while LAMP and RPA offer robust, instrument-free amplification. The choice is not necessarily either/or; the most powerful emerging applications synergistically combine RPA or LAMP for pre-amplification with CRISPR for specific detection, creating ultra-sensitive, field-deployable assays. For researchers and developers, the decision matrix hinges on the application's priority: ultimate specificity (favoring CRISPR integration), sheer speed and simplicity (favoring standalone RPA), or robust amplification of complex targets (favoring LAMP). Future directions point toward fully integrated, microfluidic 'sample-to-answer' devices, enhanced multiplexing capabilities, and quantitative digital readouts. As these technologies mature and regulatory pathways clarify, their convergence will continue to democratize precise molecular diagnostics, moving them from central labs directly to the clinic, pharmacy, and patient's bedside, revolutionizing personalized medicine and global health surveillance.